
Informed Manufacturing through Big Data Analytics of Freeze Drying Process and Equipment.
What could manufacturing look like in the future? Picture predictive analytics to minimize downtime and a control center to monitor the entire fleet of machines, i.e. a connected factory. To turn this vision into reality, a program for data analytics and health monitoring has been started at IMA Life North America in Tonawanda to provide augmented freeze dryer information by collecting and analyzing process and equipment data.
The United States Food and Drug Administration (US FDA) has issued elaborate guidance on Continued Process Verification (CPV) with regards to continual assurance for maintaining the state of a validated system. Adherence to cGMP requires a system for detecting unplanned departures from a process.
In addition to monitoring and recording data, the information collected must be reviewed to ensure the control of CQAs throughout the process.
With these guidelines as a basis, the development of advanced equipment must be complemented with a comprehensive data analytics for monitoring system health – and in this case freeze dryer system health management.
Currently, a reactionary approach is followed in the freeze drying industry. Responding to failures and events after they occur not only leads to increased downtime but also frequent scheduling changes.
Thus, while the traditional reactionary and preventive maintenance based model relies on redundancy, here we rely on data-driven decisions. Collecting discrete data from established methods during preventive maintenance does not provide specific continuous benchmarking for an equipment or process. The need to deploy a data analytics and health monitoring package for freeze dryer system management is born out of the gaps and drawbacks of the current industry standard.
1. Data Analytics and Health Monitoring at IMA Life
At IMA Life Tonawanda, a move towards establishing models from historical and real-time analysis of freeze drying data is being accomplished. A typical freeze dryer has many different subsystems, each having their own characteristic behaviors, yet working together to maintain the intricate conditions required for the drug product. The goal of this program is to create a framework for extracting useful process/systems information and implement models based on equipment, process and product knowledge to enhance production efficiency and product quality.
 Keeping the objectives of minimizing downtime and moving towards a predict and schedule model, the program proceeds in three broad phases,
Keeping the objectives of minimizing downtime and moving towards a predict and schedule model, the program proceeds in three broad phases,
• Phase I – Historical Data Analysis: To learn equipment and process specific behaviors
• Phase II – Model Building: Use algorithms to develop freeze dryer specific models
• Phase III – Sensor Network: Implement additional sensors and real-time data collection modules.
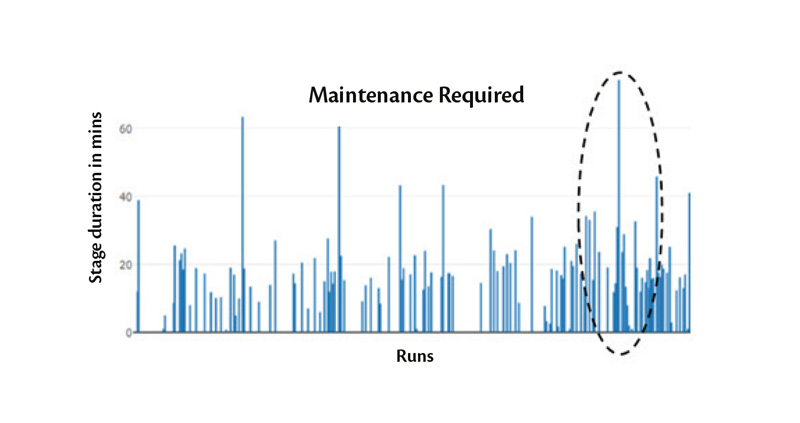
To visualize the impact of this implementation, let’s consider a classic example – evacuation of a freeze drying chamber.
The performance of the freeze dryer during the vacuum pulldown phase can be used as an indicator for leaks and health of the vacuum pump. An increasing trend in time required to evacuate a chamber to the set pressure may be a cause for concern. By monitoring the rate by which evacuation time rises, alarm limits can be implemented and notifications sent to production personnel for scheduling of predictive maintenance.
2. Looking to the Future: Lab4Life Implemen-tation
IMA Life Tonawanda is partnering with 3 major pharmaceutical customers to implement an early adoption of the data analytics and health monitoring program on production freeze dryers at their facilities.
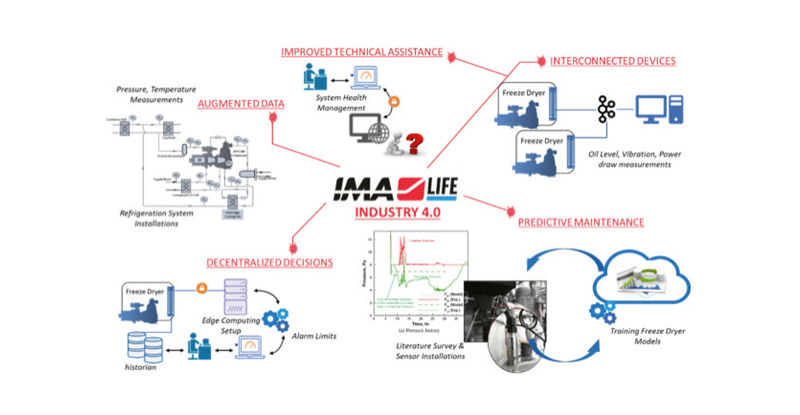
This project involves processing of over 1 years’ worth of production data, each with 0.5 million data points from about 100 different PLC tags, using an algorithm for freeze dryer signature identification and golden batch generation.
This co-development phase presents excellent opportunities for working with real pharmaceutical processing data to conceptualize, develop and test for freeze drying 4.0, towards informed manufacturing. This application ties well in IMA Digital’s portfolio for smart manufacturing and improving production efficiencies.
Paper sections:
Last Submitted Papers
- LYnfinity is just the drop away
- LYnfinity – Drying Technology
- Today, freeze dryer can use a 100% green refrigeration system, using air as the refrigeration fluid.
- DSMC simulations of vapor flow and ice dynamics in a freeze dryer condenser.
- Freeze-Drying Process Optimization.
- Increasing Vial to Vial Homogeneity: An Analysis of VERISEQ® Nucleation on Production Scale Freeze Dryers.
- Lynfinity – Freezing
- Lynfinity – Spraying
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website

