
Quick access to IMA Sectors:
Pharma | Food & Dairy | Confectionery | Tea & Beverage | Coffee | Personal & Home Care | Tissue & Nonwoven | Automation | Tobacco | E-Commerce |Nebula, high-speed decontamination tunnel for RTU components.
1. Executive summary
Part of the IMA Group, leading global manufacturer of processing and packaging machinery for the pharmaceutical and biotechnology industries, IMA LIFE specializes in advanced aseptic processing, freeze-drying, and filling technologies, and creates innovative solutions that aim at achieving the highest levels of safety and efficiency in pharmaceutical production.
IMA LIFE recently introduced NEBULA, a new technological solution for the introduction of RTU (ready-to-use) pre-sterilized material into an isolated aseptic environment (Grade A area) through surface decontamination methods. The goal of technologies such as NEBULA is to meet stringent regulatory requirements, supporting the development and manufacturing of high-value pharmaceutical products.
This white paper examines the technology at the heart of NEBULA and the benefits it will ensure for safer, more efficient aseptic processing lines.
2. State of the art
The loading process of sterile primary containers (vials/syringes/carpules) or sterile materials into filling systems for injectables or other types of parenteral drugs has historically been one of the most critical aspects of the entire fill-finish process. In the case of bulk containers, the established approach of introducing the containers using washers and depyrogenation tunnels is still predominant and well-established in aseptic plants.
At the same time, the sector of primary containers managed as pre-sterilized materials is quickly expanding. Primary containers can be transported within the Grade A area through specific protective packaging, commonly referred to as “RTU tubs” or “RTU trays”. RTU tubs are equipped with primary containers inserted into “nests” and they are used for vials, syringes, and cartridges (ref. ISO 11040-7 “Packaging systems for sterilized subassembled syringes ready for filling”). Trays are similar but are typically dedicated to vials and do not have nests for separating the containers.
Tubs and trays are specialized containers designed for the transportation of primary containers in aseptic isolators. They play a crucial role in maintaining the sterility of the contents during handling and transfer processes.

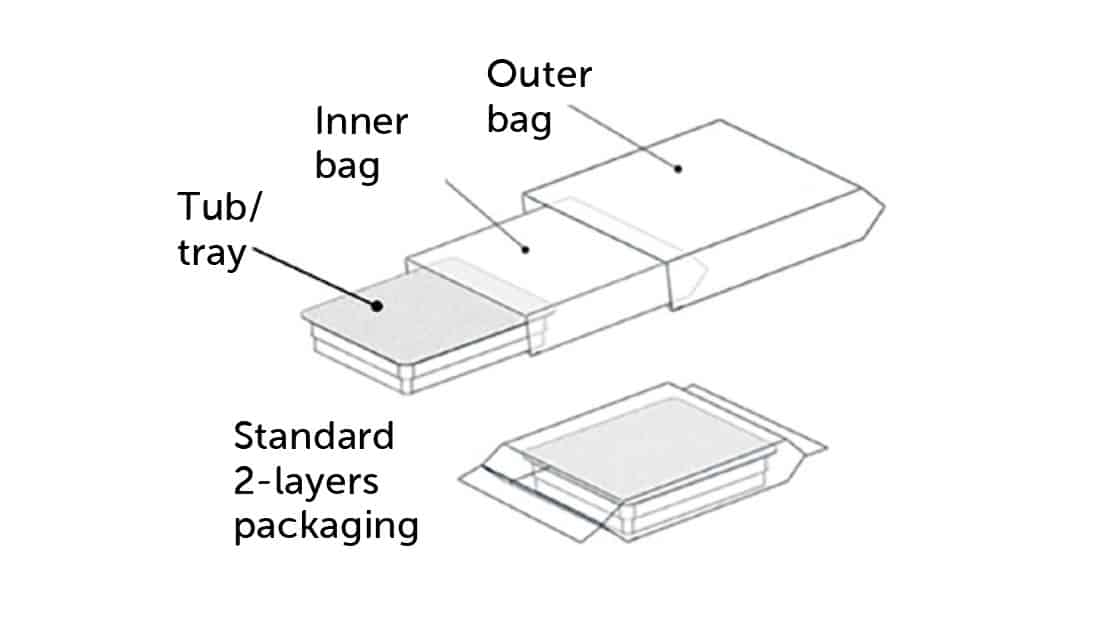
Both tubs and trays are essential in aseptic processing, as they provide a sterile barrier and support effective workflow within the Grade A area, ensuring that the primary containers remain uncontaminated until they are ready for use in pharmaceutical manufacturing or laboratory procedures.
As described in GMP guidelines and the literature on aseptic processes, there are two primary methods for introducing RTU (ready-to-use) materials into an isolator while ensuring the sterility of the Grade A environment and the packaging.
- Surface decontamination: This approach focuses on reducing the bioburden on the external surfaces of RTU materials before they enter the isolator. Automated decontamination can be achieved using technologies such as Vapor Phase Hydrogen Peroxide (VPHP), electron beam, or UV pulsed light. These methods effectively decontaminate the outer surfaces of materials to prevent contamination of the critical area.
- Mechanized de-bagging with No-Touch-Transfer (NTT): This method involves a mechanized process to remove the external packaging without human contact, using the No-Touch-Transfer (NTT) technique. NTT ensures that the sterile contents are introduced into the Grade A area without any risk of contamination from handling, maintaining the integrity of the aseptic environment.

There are different types of surface decontamination technologies, here a list of the most common:
- VPHP decontamination using large/small transfer chamber
- Pulsed light decontamination
- Electron beam (E-beam) decontamination
From the perspective of No-Touch-Transfer systems, there are solutions at various levels of automation:
- Completely manual solution, where the operation is fully performed by operators in the clean room.
- Semi-automatic solution, where the unbagging and introduction operations are assisted by an automation system, but still managed by an operator.
- Automatic solution, where the unbagging and introduction operations are entirely delegated to automatic or robotic solutions.
These current technologies are characterized by their own advantages and disadvantages. IMA LIFE’s proposal aims to overcome the current limitations of existing technologies.
3. Technology and regulatory challenges
The primary requirement of No-Touch-Transfer (NTT) systems is that the sterile condition of the primary packaging is ensured. The process of unsealing (peeling) tubs or trays must occur in a Grade A environment, with the inner bag’s sterility being crucial to avoid contamination risks within the Grade A area. Additionally, the packaging type, whether single bag, double bag, or vacuum-sealed, can significantly influence the design and performance of automated systems. While automation enhances sterility assurance, it also imposes considerable constraints on the layout and complexity of production lines. Furthermore, the responsibility for guaranteeing packaging sterility, including the inner bag, lies with the tub and tray suppliers. However, not all suppliers can reliably ensure the sterility and final integrity of the inner bag, presenting a critical regulatory challenge for the industry.
The key considerations for managing ready-to-use (RTU) components with a No-Touch-Transfer (NTT) approach concern three main stages: RTU production site guarantees, transport to the production site, and incoming preparation for the aseptic process. At the production site, it is crucial to have comprehensive knowledge of the packaging process and materials, ensure supplier sterilization and qualification before release, and clearly define the sterile barrier’s limits – whether inside or outside the tub/tray. During transport, maintaining RTU integrity through proper logistics and shipment handling is essential. Once in the aseptic processing area, integrity checks (questioning whether visual inspection is sufficient) and routine quality checks on RTU samples must be performed. Standard Operating Procedures (SOPs) are also necessary for the cleaning and sanitization of RTU containers before processing, as well as for its transfer and handling within the cleanroom environment.
The combination of these critical issues results in a process that is not entirely free from risks, which can only be partially mitigated and are still subject to regulatory compromises. By design, it is not possible to guarantee the sterility of the inner bag, which therefore allows potentially contaminated material to enter the Grade A environment. Moving to surface decontamination technologies, it is possible to summarize the pros and cons of different technologies.
1. VHP Decontamination using large/small transfer chambers
Advantages:
- Possibility to manage different types of packaging
- Small or large loads possible
- Validation up to 6-log microbiological reduction
- Standard VPHP technology
- Room temperature process
Limitations:
- Not suitable for fast RTU loading (up to 5/6 containers per minute requested by high-speed filling lines) due to batch processing time
- Risks of H2O2 residues on primary container
- Requalification in case of load or RTUs change
- Complex automation request for chamber loading-unloading (usually robotic assisted)
2. Pulsed light decontamination
Advantages:
- Faster batch decontamination
- No chemical or ionizing radiation involved
- No chemical residues
Limitations:
- Difficult to validate 6-log
- Sensitive to shadowing effects
- Need of robotic assistance to achieve complete decontamination
- Light pulses are fast, but multiple shots and robotic manipulation requested can decrease the process speed
- Short lifetime of pulsed light source
- Specific BIs, using different specific spores and a “light permeable” transparent packaging,used in CD and PQ
3. Electron beam (E-beam) decontamination
Advantages:
- Short decontamination time
- Suitable for high-speed RTU introduction (high-speed filling lines)
- Consolidated technology
Limitations:
- High capex and operational costs
- Risks due to ozone generation and residues on the primary container Ionizing radiationand related risks for operator and the environment (lead shielding required and HSE concerns)
- Short lifetime of E-beam sources
- Specific validation based on absorbed dose, usually not BI assisted.
As a final consideration, the current surface decontamination technologies and No-Touch Transfer approaches present several limitations. The application of these well-established technologies still faces operational and regulatory limitations that have not yet been fully overcome. In response to these challenges, IMA LIFE has identified an innovative solution that simplifies and strengthens surface decontamination processes, offering a more robust and efficient approach.
4. Nebula: the continuous decontamination tunnel approach
The primary objective was to replicate the strategic advantages of an E-beam tunnel by utilizing traditional hydrogen peroxide-based decontamination methods. IMA LIFE has successfully adapted an existing technology from food processing, where high-concentration vaporized hydrogen peroxide (VPHP) exceeding 8000 PPM is currently employed for the decontamination of bottles and pouches. By optimizing the combination of temperature, high concentration, and airflow distribution, it becomes feasible to achieve the 6-log decontamination in approximately 30 seconds.
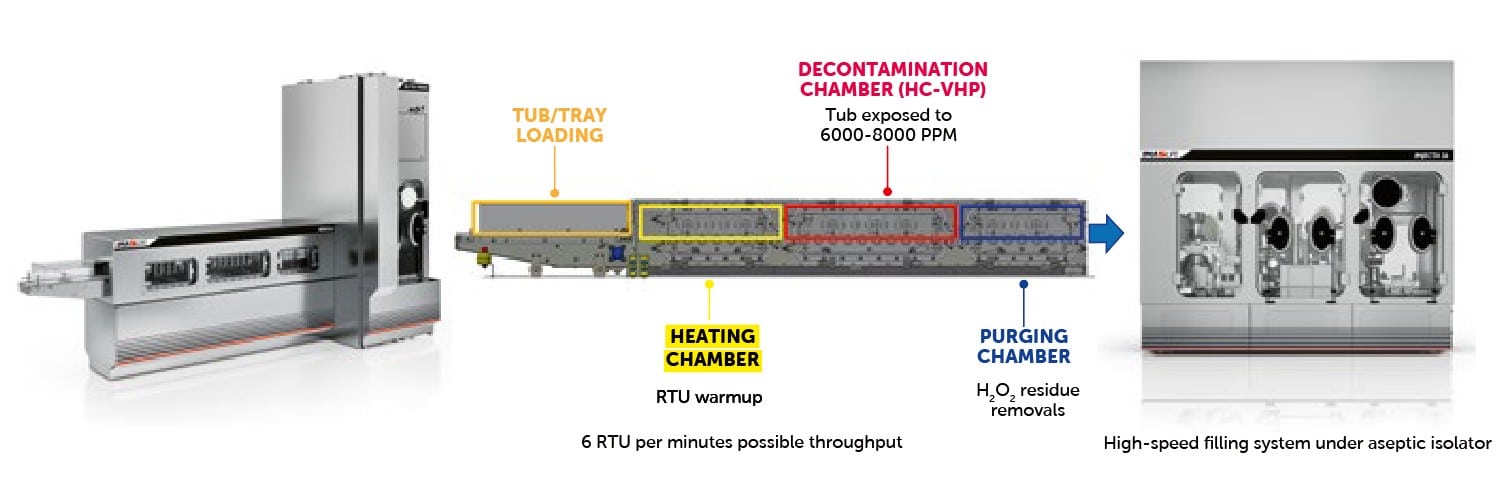
The process is divided into three chambers, maintained at different pressures to achieve dynamic segregation of the VPHP within the machine, allowing for an ‘open’ process without a physical airtight separation between the decontaminated area and the external cleanroom environment. This approach enables continuous operation of the decontamination tunnel, where the RTU container passes through the three chambers, moved by a simple roller-based conveyor system.
Through diffusion rings placed along the path, the tub or tray is first exposed to hot, dry air to increase its surface temperature, then to high-concentration VHP at a temperature between 70°C and 75°C, and finally again to hot, dry air to promote the desorption of hydrogen peroxide residues.

The final result is an RTU container that is decontaminated on the outer surface, demonstrating a 6-log reduction while avoiding hazardous internal H₂O₂ residues. The process can be performed continuously, allowing multiple containers to enter the tunnel simultaneously, enabling a system capable of introducing up to 5/6 tubs/trays per minute into the aseptic area in a compact space. The system employs various technological solutions, including high-prevalence blowers for generating the flow rates required for the pressurization of the treatment chambers. It also utilizes absolute 0.22 µm grade filters for process air filtration within the tunnel and a standard hydrogen peroxide solution at 35% concentration.
The tunnel is initially fully decontaminated (6-log standard initial VPHP cycle) before entering production, ensuring that the tunnel chamber environments are fully aseptic. These chambers are equipped with Grade A air supply and maintain a tightness level comparable to standard aseptic isolators.
Automatic shutter systems between the tunnel chambers are one key feature to maintain the pressure cascades. The chamber ventilation is designed to prevent cross-contamination of H₂O₂. Dynamic containment of H₂O₂ is enhanced through computational fluid dynamics (CFD) and a patented design. Additionally, specific decontamination cycles and consequent PQ are implemented for each type of RTU container.
5. Advantages of Nebula technology
The advantages of this new technology are here summarized:
- Robust surface decontamination: The system uses high-concentration vaporized hydrogen peroxide (VPHP) at over 8000 PPM, combined with optimized temperature and airflow, to achieve a 6-log decontamination in about 30 seconds, ensuring rapid and effective surface decontamination and avoiding residual risks of No-Touch-Transfer solutions.
- Continuous operation: The tunnel design enables continuous decontamination of multiple containers simultaneously, with up to 5/6 tubs/trays entering the aseptic area per minute, significantly increasing throughput and efficiency versus current traditional VPHP methods.
- Dynamic chamber segregation: The process involves three pressurized chambers that dynamically segregate VPHP without requiring airtight physical barriers, allowing an ‘open’ process and smooth material flow between the decontaminated and cleanroom environments.
- Advanced airflow and CFD design: Diffusion rings and computational fluid dynamics (CFD) optimize VPHP distribution and containment, ensuring uniform decontamination across surfaces and minimizing the risk of hydrogen peroxide residue.
- Simple and compact system: The system is designed to be connected to fill-finish aseptic lines similarly to e-beam tunnels. The layout impact is lower than a traditional No-Touch-Transfer automated system, simplifying handling and avoiding the management of double-bagged packaging, with a reduced risk of jams and machine downtime.
In conclusion, the system achieves the advantages of more complex technologies, such as high-speed RTU handling in smaller spaces, with reduced mechanical complexity, lower HSE risks, and lower RTU packaging requirement (single bags protected RTU instead of double bags), making it easier to apply surface decontamination processes to aseptic filling lines.
6. Conclusion
The technology is being promoted principally with an eye to improving process efficiency and process safety for pharmaceutical companies specialized in the injectable drug manufacturing sector. An extensive amount of data generated over a full year has been collected from a prototype installed inside a laboratiry environment. Valuable information regarding the technology’s microbiological reduction capability has been gathered and potential H₂O₂ residues inside primary containers have been fully assessed.
Various types of packaging have been analyzed, and the solution is currently available on the market in combination with robotic filling systems for RTU containers, typically high-speed lines for PFS or combo lines for vials and syringes.

