
Quick access to IMA Sectors:
Pharma | Food & Dairy | Confectionery | Tea & Beverage | Coffee | Personal & Home Care | Tissue & Nonwoven | Automation | Tobacco | E-Commerce |Minitablets made into hard capsules or administered using special dosing units, as well as pellets in hard capsules or compressed into tablets, offer the advantages of multiparticulate drug delivery systems and are suitable for controlled drug release using polymer coatings.
Comparison of minitablets and pellets as multiparticulate drug delivery systems for controlled drug release.
|
|
Abstract
Minitablets made into hard capsules or administered using special dosing units, as well as pellets in hard capsules or compressed into tablets, offer the advant ages of multiparticulate drug delivery systems and are suitable for controlled drug release using polymer coatings. Four different kinds of solid drug preparations were manufactured and investigated concerning drug release. Inert pellets were coated with the model drug sodium benzoate and, in a second step, with the insoluble polymer ethylcellulose. The coated pellets were compressed into minitablets and into normal tablets.
Another kind of minitablet was compressed from a sodium benzoate compression mixture and finally coated with ethylcellulose. The coating of the tablets was performed using fluidized bed technology. The sodium benzoate release plots of the coated pellets show a lag time and retarded release according first-order kinetics. The minitablets and normal tablets compressed from pellets release sodium benzoate according to first-order kinetics as well, but without the lag time due to distinct ethylcellulose layer destruction during tableting. The release is retarded with increasing ethylcellulose layer thickness on directly compressed minitablets. The different formulations of coated pellets, minitablets, and normal tablets offer a broad choice for variable drug release kinetics depending on the biopharmaceutical and pharmacological requirements.
1. Introduction
In contrast to classical single-drug units like tablets, hard and soft gelatine capsules, and single powder or granule doses in sachets and stick packs, minitablets and pellets are small, solid drug units providing several distinct advantages, i.e., dose definition through the combination of single units, and homogeneous spreading after administration in the stomach and small intestine, therefore increasing the adsorption area and improving bioavailability.
Tablets developed in a solid drug dosage form for human use are in the range of 4–15 mm in diameter, whereas minitablets range from 1 to 4 mm, making them suitable for the manufacture of multiparticulate drug delivery systems. Minitablets may be filled into hard gelatine capsules or compressed into tablets as the final drug formulation. There has been an increasing interest in minitablets over the last 20 years. Until now, the investigation of minitablets has focused on their compression behavior and properties. Minitablets are compressed using lower compression force than classical tablets and exhibit lower tensile strength, but also exhibit sufficiently low friability depending on the formulation components. The capping tendency is reduced in some cases compared to large tablets,[1] and doses of up to 90% of some critical drug substances may be successfully compressed.[2] The content uniformity of minitablets depends on the component particle size, particle size distribution, the high level of interactive blending of the components,[3] compression blend flow properties,[4] and the drug substance’s physical behavior.[5] Controlled drug release is one of the most important aims of minitablet manufacturing. In analogy to large tablets with slow and sustained release, matrix minitablets were investigated that consisted of insoluble, but slowly swellable ethylcellulose,[6] and high-molecular hydroxypropylmethylcellulose,[7-9] polyvinylacetate/polyvinylpyrrolidone,[10] and pHvalue-sensitive acrylate polymers.[11]
Similar insoluble acrylate polymers,[12,13] copolymer vinylpyrrolidone-vinylacetate,[14] ethylcellulose,[15] and hydroxypropylmethylcellulose acetate succinate,[9] were used as coating materials for minitablets with sustained drug release, distinct lag time, and enteric coating properties. Minitablets were developed with different application and indication aims. Mucoadhesive minitablets for ocular medication consist of high-molecular polyacrylic acid or waxy maize starch.[16,17] After administration into the saccus conjunctivae, the minitablets swell, forming a gel that sticks to the mucosa and releases the drug over a long time period. For the prolongation of the gastric emptying time, the minitablets were compressed into large tablets with effervescent substances for gas generation and a larger content of high-molecular hydroxypropylmethylcellulose, leading to a prolonged residence time in the stomach and sustained drug release.[18–20] The absorption of peptides after oral administration was improved and delayed by using N-trimethyl chitosan chloride as the main component of minitablets.[21] Drug release was optimized through drug complexation with ß-cyclodextrin.[22] Minitablets have also received increasing interest in terms of dose-controlled use for children. The taste of sodium benzoate was successfully improved by the use of hard fat in minitablets,[23] and the suitability of powdered milk for the production of paracetamol minitablets was investigated.[24] Compared to minitablets, pellets and coated pellets are used for a longer time as the drug-containing components of multiparticulate drug delivery systems. Research and development in the field of pharmaceutical pellets include processing, i.e., extrusion/spheronization,[25] fluidized bed rotor agglomeration,[26] and layering and coating with enteric,[27,28] and retarding polymers like ethylcellulose.[29-33] Drug substances may be incorporated into the pellets, layered onto the surface of the pellets, or both for the initial and maintenance dosage.[34,35] Minitablets and pellets are comparable so far, but differ in size, with drug-loaded pellets for pharmaceutical use being in the range of 100–800 μm. The contents of minitablets and drug pellets are filled into hard capsules or compressed into tablets. Consequently, some authors compare the suitability and the properties of pellets and minitablets with the same drug substance and similar formulations, indicating the advantages and disadvantages of both dosage forms. Drug release depends on the kind of coating polymer and layer thickness, the wetting and water absorption of the neutral cores, and formulation changes induced by matrix formers.[36] The release of easily soluble drug substances from ethylcellulose-coated pellets according to the manufacturing method, kind of binder used in the wet granulation, and layer thickness increased in comparison to minitablets due to their significantly higher specific surface area.[37] For prolonged release, pellets require a much thicker coating layer compared to minitablets. The coating layer thickness of similarly formuted minitablets and pellets was successful measured using dynamic image analysis.[38] The use of minitablets competes with new development dosage forms and modifications for sustained release.[39,40] The aim of the present paper was to examine the development of coated pellets and minitablets and to compare the different solid dosage forms on the release kinetics of easily soluble sodium benzoate as a model drug substance. Therefore, three different solid dosage forms were manufactured. Inert microcrystalline cellulose pellets were used as starter cores. In a first step, the pellets were coated with sodium benzoate, and in a second step, with the ethylcellulose as an insoluble retarding polymer (EC pellets). The coated pellets were compressed into minitablets type 1 (MT1) and normal tablets (NT). Furthermore, minitablets type 2 (MT2) were manufactured through the direct compression of sodium benzoate and excipients (filling agent, disintegration agent, lubricant) and were then coated with ethylcellulose (Figure 1). The coating was performed using fluidized bed technology. The influence of the process parameters and formulation parameters on the coating was evaluated, as well as the impact of the compression procedures on the product properties and sodium benzoate release. Finally, the products were compared regarding the product parameters and benefits for industrial manufacturing.

Figure 1: overview of three different solid dosage forms for sustained release.
 |
 |
2. Materials and methods
2.1. Materials
All excipients were used as received. The quality of all substances refers to pharmacopoeia [Ph. Eur. 2020]. Inert microcrystalline cellulose pellets (Cellets®200, IPC, Dresden, Germany), sodium benzoate (Chemie Vertrieb, Magdeburg, Germany), polyvinylpyrrolidone (PVP, Kollidon 30®, BASF, Ludwigshafen, Germany), talcum (Chemie Vertrieb, Magdeburg, Germany), ethylcellulose (Surelease®, Colorcon, Dartford, UK), microcrystalline cellulose (MCC, VIVAPUR®102, JRS Pharma, Rosenberg, Germany), lactose monohydrate (Meggle, Wasserburg, Germany), high-dispersed silicon dioxide (Aerosil®200, Evonik Industries, Germany), carboxymethylcellulose, sodium salt, magnesium stearate, and zinc stearate (all substances from Merck, Darmstadt, Germany) were used.
The formulation of the sodium-benzoate-layered and ethylcellulose-coated pellets is summarized in Table 1, and the formulations of the minitablets MT1 and MT2 and normal tablets (NT) are presented in Table 2.
| Components of pellet lot | Content (%) |
| Microcrystalline cellulose (Cellets®200) | 23.2 |
| Sodium benzoate | 30.2 |
| Polyvinylpyrrolidone | 1.6 |
| Talcum | 0.6 |
| Ethylcellulose | 44.4 |
| Total | 100.0 |
Table 1: formulation of the final pellets coated with sodium benzoate and ethylcellulose.
In the case of the minitablets MT1, polyvinylpyrrolidone was added to the sodium benzoate layering fluid to increase the plasticity and mechanical stability of the sodium benzoate layer, and talcum was added to prevent the pellets from sticking during the process (Table 2). Surelease® is a ready-to-use aqueous coating dispersion made of ethylcellulose and the addition of further excipients is not necessary. Polyvinylpyrrolidone and talcum were not included in the MT2 minitablet lot due to the direct compression of all components. Lactose monohydrate serves as a filling agent, sodium carboxymethylcellulose as a disintegrant, magnesium stearate as a lubricant, and high-dispersed silicon dioxide as a flow promotor in the compression powder mixture in order to obtain successful compression and final tablets of high quality.
| Components of minitablets | MT1/NT content (%) |
MT2 content (%) |
| Microcrystalline cellulose | 36.6 | 38.8 |
| Sodium benzoate | 15.1 | 15.5 |
| Polyvinylpyrrolidone | 0.8 | |
| Talcum | 0.3 | |
| Ethylcellulose | 22.2 | 20.0 |
| Lactose monohydrate | 18.5 | 19.0 |
| Carboxymethylcellulose, sodium salt | 5.0 | 5.2 |
| Magnesium stearate | 1.0 | 1.0 |
| High dispersed silicon dioxide | 0.5 | 0.5 |
| Total | 100.0 | 100.0 |
Table 2: formulation minitablets MT1 and MT2 and normal tablets NT
2.2. Methods
2.2.1. Sodium benzoate layering of pellets and ethylcellulose coating in the fluidized bed
The fluidized bed layering of the inert pellets with sodium benzoate and the coating of the sodium benzoate pellets and minitablets with the ethylcellulose was performed using a lab-scale fluidized bed coater (Mylab, IMA S.p.A., Bologna, Italy) equipped with the central partition and the bottom spray setup. The fluidized bed coating process parameters are reported in Table 3.
| Process parameters | m.u. | Sodium benzoate layering |
EC pellets |
MT2 ethylcellulose coating |
| Batch size | kg | 3.0 |
1.6 |
2.0 |
| Process air rate | m3/h | 125 | 125 | 200 |
| Process air temperature | °C | 70 | 60 | 70 |
| Product temperature | °C | 43 | 41 | 55 |
| Spray pressure | bar | 2.0 | 2.0 | 2.0 |
| Spray rate | g/min | 30 | 15 | 10 |
| Spray nozzle diameter | mm | 1 | 1 | 1 |
Table 3: fluidized bed process parameters, sodium benzoate layering, and ethylcellulose coating of pellets and minitablets.
The direct-compressed minitablets MT2 were coated with ethylcellulose using an increased process air rate of 200 m3/h compared to the sodium benzoate layering and the ethylcellulose coating of pellets (125 m3/h, Table 3). Therefore, the water evaporation rate was increased and the product temperature was raised to 55 °C compared to 42 °C for MT1 and NT. The spray rate was lower in all cases of ethylcellulose coating compared to sodium benzoate layering to prevent the pellets and minitablets from sticking and to stop the fluidized bed from over-wetting and breaking down.
2.2.2. Tableting
The Prexima 80 (IMA S.p.A., Bologna, Italy) rotary tablet press was used for the minitablet compression (MT1 and MT2). It was equipped with a mixed turret EU-B and EU-D, suitable for laboratory trials, consisting of EU-D dies and EU-D biconvex round multitip punches of 2 mm in diameter (24 tips per punch). The normal biplane tablets (NT) of a 6 mm diameter round shape were compressed using the same press and tablet press configuration.
Two levels of compression forces were used for the minitablets (6 and 11 kN) and normal tablets (11 and 31 kN) with the aim of assessing the release profile of the solid oral dosage forms. The thickness of the minitablets was 3 mm and that of the normal tablets was 4 mm.
2.2.3. In vitro release of sodium benzoate
An in vitro release investigation was carried out using a dissolution tester (PTW II, Pharma Test, Hainburg, Germany) equipped with 6 vessels and paddle at a temperature of 37 °C and a rotation rate of 50 rpm in purified water (1000 mL), according to the pharmacopoeia method [Ph. Eur. 2020]. A suitable measure of EC pellets (33 mg) or number of minitablets (10 and 12 for MT1 andMT2, respectively), amounting to approximately 10 mg sodium benzoate, was placed into each vessel. The samples of the release fluid (10 mL) were withdrawn at predetermined time intervals over at least 120 min and were filtered using a membrane filter of pore size 0.22 μm. The sodium benzoate content of the filtrate was directly measured using UV spectroscopy. The sodium benzoate content of the EC pellets and minitablets was measured using UV spectroscopy (Spekol 1300, Analytik Jena, Jena, Germany) at 220 nm wavelength and 10 mm quartzite cuvette length, compared to blind purified water. Therefore, a defined measure of EC pellets or minitablets amounting to 10–12 mg sodium benzoate was dispersed in purified water, agitated for 30 min, and filtered, and then the filtrate was transferred to a 1 L volumetric flask and filled up to the measurement line.
2.2.4. Scanning Electron Microscopy (SEM)
The morphology of the pellet and the minitablet surfaces was examined using SEM (Type PWb6703/SEM 515, Philips Corp., Eindhoven, The Netherlands). Accordingly, the samples were coated with gold–palladium plasm for 230 s under an argon atmosphere (SCD 040, Balzers Union, Liechtenstein).
2.2.5. Powder characterization
The particle size, size distribution, and sphericity of the pellet products were measured using dynamic image analysis (Camsizer® P4, Retsch, Haan, Germany). The bulk density of the pellet products (100 g) was measured using an SVM 102 tap volumeter (Erweka, Heusenstamm, Germany) with a 250 mL graduated cylinder. The tablet friability was investigated using a TA 10 friabilator (Erweka, Heusenstamm, Germany). In this process, a 6.5 g weight of the tablets is exposed to friction and fall forces over 4 min using a rotating wheel 30 cm in diameter with a baffle. The tablets are inspected for the percentage mass lost through chipping. The Ph. Eur. Specification allows maximum of 1% mass to be lost [Ph. Eur. 2020].
2.2.6. Linearization of release curves
The evaluation of the release curves was performed according to different models of release kinetics used by several authors.[41–44] In the first step of release evaluation, the amount of a cumulatively released substance is plotted compared to time. Linear curves arise in the case of zero-order kinetics, i.e., equal drug amounts are released at equal time intervals (Equation 1). This behavior is typical for long-acting drug delivery systems with pronounced retarded release like implants, oral therapeutic systems in the sense of an osmotic pump, ocular therapeutic systems, and special kinds of microparticles and nanoparticles with biodegradable polymers, and is not typical in the case of pellets and tablets.
![]()
First-order release kinetics is typical for slightly soluble drugs in solid preparations like tablets, pellets, and granules dominated by slow dissolution and diffusion control.
The dissolution, or the release rate, is highest at the beginning according to the large concentration gradient — as the most important factor in Fick’s first law for transport flow density by diffusion (Equation 2) — and diminishes with process time.
![]()
The released drug amount at moment t is calculated using Equation 3, and linearization gives the Sigma Minus function (Equation 4).
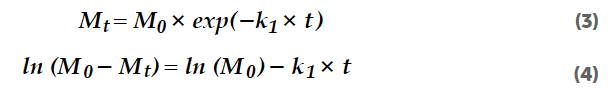
Another approach to the first-order kinetics evaluation of release processes includes the RRSB function (Equation 5) and linearized form (Equation 6).
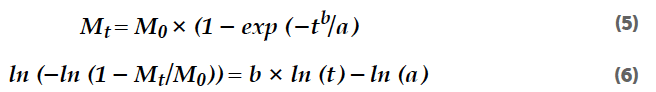
The square root function (Equation 7) is preferred in cases of the diffusive transport processes of non-disintegrating matrices and semisolid systems like ointments and gels.
![]()
The difference factor describes the relative error between two release profiles calculated from the cumulative released amounts R and T at distinct moments for reference and test formulation (f1, Equation 8). T tor is based on the sum of the of the released drug a mounts (f2, Equation 9) of the reference and test formulation, and takes a value of 100 in the case of identical release profiles and values between 50 and 100 for similar release profiles. Both factors are used for the comparison of generics and standard drug product release rates. In this study, the factors are used to evaluate the similarity (or difference) of the sodium benzoate release from minitablets, normal tablets, and EC pellets.
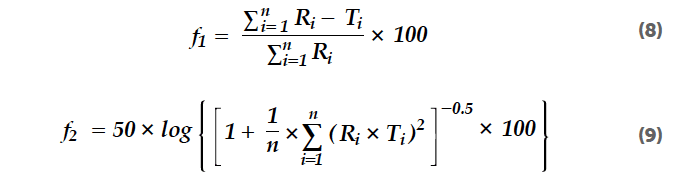
3. Results and discussion
3.1. Manufacturing of coated pellets and tableting and coating of tablets
The process of consecutive Cellets®200 layering with sodium benzoate and ethylcellulose coating in the fluidized bed was performed according to earlier investigations.[34]
The ethylcellulose coating of MT2 was accompanied by agglomeration due to the greater contact area of the minitablets in comparison to the pellets. Successful agglomeration mitigation was achieved by increasing the process air rate and reducing the spray rate from 15 to 10 g/min (Table 3). During the ethylcellulose coating of MT2, the samples of intermediate product were taken at 10% and 20% ethylcellulose content in order to investigate the sodium benzoate release.
3.2. Product properties
3.2.1. EC pellets
The ethylcellulose-coated sodium benzoate pellets provide the advantage of a multiparticulate drug delivery system with a median x50.3 of 574 μm. The bulk density of 0.74 g/mL hints to the slight densification and physical stability of the pellets. The surface is smooth and homogeneous with only low roughness (Figure 2). The inner microcrystalline cellulose pellet is surrounded by layers of sodium benzoate and ethylcellulose and the pellets indicate high sphericity at 0.96.
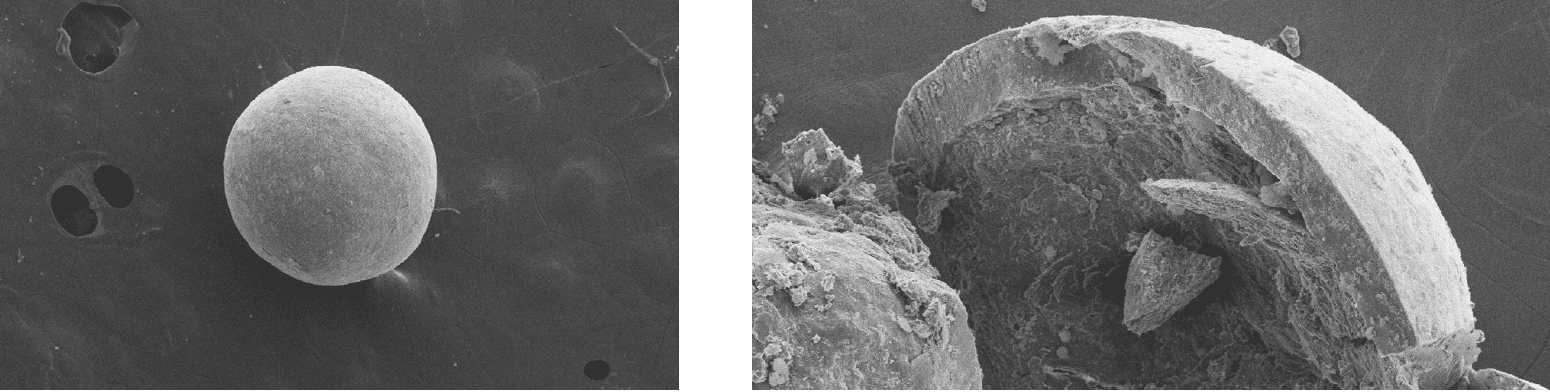
Figure 2: SEM of Cellets®200 layered with sodium benzoate and ethylcellulose: complete pellet (left) and section with core and coating layer (right).
3.2.2. Minitablets and normal tablets
The minitablets MT1 and MT2 (before ethylcellulose coating) show relatively high thickness (3 mm, Table 4) compared to diameter (2 mm). MT1 contains 50% ω/ω EC pellets and MT2 is compressed from a powder mixture lacking in polyvinylpyrrolidone and talcum and, finally, coated with ethylcellulose. The increased average weight of MT1 (8.46 mg) compared to MT2 (7.36 mg) results from the high density of the EC pellets in MT1. The sufficient hardness and mechanical stability of all three kinds of tablets are evidenced by their low friability (Table 4).
| Samples | Avg. weight (mg) | Weight uniformity (%) | Diameter (mm) |
Thikness (mm) |
Friability (%) |
| MT1 | 8.46 | 4 | 2 | 3 | 0.5 |
| MT2 | 7.36 | 4 | 2 | 3 | 0.0 |
| NT | 151.8 | 1 | 6 | 3 | 0.1 |
Table 4: properties of minitablets MT1 and MT2 with ethylcellulose layer and normal tablets NT.
The SEM microphotographs of MT1 compressed from EC pellets show the threedimensional dispersion of the pellets (Figure 3).

Figure 3: SEM of minitablets MT1 compressed from EC pellets and excipients: complete minitablet (left, middle) and section (right).
The interparticulate space between the pellets is filled with the mixture of powder excipients (Table 2). The pellets are visible not only throught SEM imaging, but also using stereo light microscopy.
The ethylcellulose-coated MT2 minitablets manufactured from the powder mixture and not from the pellets show a smooth surface (Figure 4). The section indicates the minitablet’s core of compressed powder components surrounded by ethylcellulose film.

Figure 4: SEM of minitablets MT2 compressed from powder mixture of sodium benzoate and excipients and coated with athycellulose: complete (left) and section (right).
3.3. Sodium benzoate release from EC pellets, minitablets MT1, and normal tablets NT
3.3.1. Release versus time, zero-order kinetics
The ascents of the MT1, 11 kN and NT, 31 kN release plots are similar, but the plot of the pellets is increased (Figure 5, double linear plot).
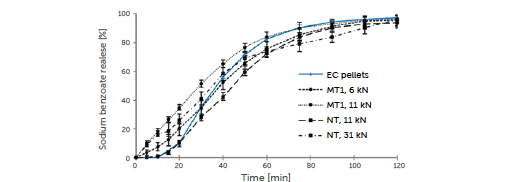
Figure 5: sodium benzoate release from EC pellets (blue line), minitablets MT1 compressed with compression force 6 and 11 kN, and normal tablets NT compressed with 11 and 31 kN (black lines) versus time.
The sodium benzoate is released at a high rate after a lag time and there is obvious sufficient wetting and swelling of the ethylcellulose film. The ethylcellulose-coated pellets release the sodium benzoate with a lag time of 10 min due to the delayed wetting of the homogeneous surface consisting of insoluble ethylcellulose and the slow swelling of the polymer (Figure 5). After a lag time of 10 min, the release is somewhat increased (steeper ascent) compared to the minitablets and normal tablets, resulting in similar release plots after 60 min. In all cases, the sodium benzoate release reaches 90% after 90 min. The minitablets MT1 and normal tablets NT compressed from the same EC pellets do not show a lag time. In contact with water, the tablets undergo disintegration and the liberation of the pellets, and the sodium benzoate release starts immediately, contrary to the pellets. The diminishing release rate (Figure 5) in the case of the tablets may derive from both the delayed tablet disintegration and the wetting of the pellets in the inner tablet volume or in the fragments. In addition, the compression of the EC pellets into tablets is connected with the pressure onto the pellets, and, therefore, onto the ethylcellulose layer. This is accompanied by mechanical friction, generating partial layer cracking and leading to increased release and lack of lag time. Release plots at different compression forces indicate the impact factor on the drug release. With MT1 as well as with NT, the release rate is increased under high compression force (11 kN MT1 and 31 kN NT, Figure 5). A high compression force leads to stronger destruction of the ethylcellulose layer and the pellets themselves, and to faster release. The overlapping and interaction of the simultaneous ongoing processes lead to the present release behavior. The compression of the ethylcellulose-coated pellets into minitablets MT1 does not pose any advantages in comparison to compression into normal tablets NT regarding the sustained sodium benzoate release (Figure 5). The variation in the release from minitablets is increased compared to EC pellets and normal tablets compressed from pellets. Normal tablets produced under a low compression force (NT, 11 kN) show the most similar release profile to the reference profile of pellets. As expected, the results obtained for both NT and MT1 show that the main retardation effect on the drug release profile depends more on the decrease in the compression force in comparison to the disintegration rate of the tablets. This can be explained by the preferred ethylcellulose layer breakage of the coated pellets on the external side of the cores due to the compression force of the tablet stamps.
3.3.2. Linearization of the sodium benzoate release plots of MT1, MT2, and EC pellets
The first-order kinetics of the sodium benzoate release is most obvious for MT1 compressed with a force of 11 kN and NT with a force of 31 kN, with a coefficient of determination (CoD) of 0.99 (Table 5).
| Coefficient of determination R2 | MT1 (11 kN) | NT (31 kN) | Pellets |
| Zero-order | 0.87 | 0.91 | 0.90 |
| Square root | 0.95 | 0.96 | 0.88 |
| Sigma minus plot (first-order) | 0.99 | 0.99 | 0.95 |
| RRSB (first-order) | 0.99 | 0.98 | 0.95 |
Table 5: coefficient of determination of sodium benzoate release from MT1, NT, and EC pellets.
The initial phase of the wetting and swelling of the ethylcellulose film is short, lag time does not occur, and the dissolution and the release rate are high at the beginning and decelerate in the terminal phase. The CoD in the case of the pellets amounts to only 0.95 due to the pronounced lag time at the beginning resulting from the wetting and swelling of the comparably thick and homogeneous ethylcellulose layer. The zero-order kinetics and square root show a lower CoD, appearing less probable. The Sigma Minus plots show only little deviation of the ascents (Table 6).
| Parameter | MT1 (11 kN) | NT (31 kN) | Pellets |
| Ascent Sigma Minus | 0.04 | 0.03 | 0.04 |
| RRSB: 1/a (scale) | 0.22 | 0.23 | 0.10 |
| RRSB: b (form) | 1.27 | 1.15 | 2.47 |
| RRSB: t63.2% (time in min) | 35 | 46 | 60 |
Table 6: ascent first-order release and parameters of RRSB plot: MT1, NT, and EC pellets.
The form parameter b > 1 (RRSB plot) hints to biphasic release with a slow rate in the initial phase. The scale parameter 1/a, corresponding to the release rate, is diminished for pellets (0.10) compared to tablets (0.22 and 0.23) regarding the complete release curve resulting from the lag time. Slow release corresponds to a higher time parameter (t63.2%, 60 min for EC pellets versus MT1 35 min and NT 46 min, Table 6).
3.3.3. Difference factor and similarity factor of sodium benzoate release from MT1, NT, and EC pellets
The sodium benzoate release profiles from MT1, 11 kN and NT, 31 kN are similar (difference factor 11, Table 7).
| Parameter | Evaluation | MT1/NT | MT1/NT EC pellets |
NT/EC pellets |
| Difference factor | “similar” 0–15 | 11 | 32 | 21 |
| Similarity factor | “similar” 50–100 | 77 | 68 | 73 |
Table 7: Difference factor and similarity factor for comparison of release from MT1, NT, and EC pellets.
The tablets are compressed from the same coated EC pellets. Otherwise, difference factors above 15 (MT1/EC pellets 32 and NT/EC pellets 21) indicate no similarity. The reasons are the lag time and the more homogeneous ethylcellulose film in the case of the pellets compared to the tablets. All three similarity factors are in the range of 50–100, hinting to similarity. The similarity factor is calculated based on deviation squares, and is not as sensitive as the difference factor. In this case, the influence of lag time is not as strong as in the case of the difference factor (Table 7).
3.3.4. Results for MT2 with different ethylcellulose coatings
The cores without an ethylcellulose coating (0% ethylcellulose, Figure 6) release the sodium benzoate immediately due to the absence of a diffusion polymer and the high solubility of sodium benzoate.
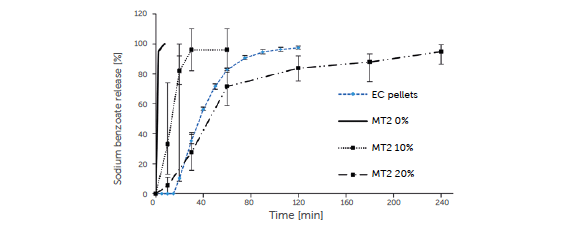
Figure 6: sodium benzoate release from EC pellets (blue line), from uncoated minitablets MT2 (solid line), and from MT2 with increasing amounts of ethylcellulose (10%, dotted line and 20%, dashed line) versus time.
A distinct retarded release is observed for MT2 with a 10% ethylcellulose coating, but without a certain lag time. The release from MT2 with a 20% ethylcellulose coating is similar to the EC pellets with a high ethylcellulose content at 44.4% (Figure 6). One should expect a lower release rate for MT2 due to a somewhat thicker ethylcellulose layer as a result of the lower surface area compared to EC pellets. The reason for the similar release is explained by the thinner coating layer at the edges of the minitablets. This is a critical point compared to the nearly ideal spherical pellets. In the case of a comparable thin ethylcellulose layer, the film will erupt preferentially at the edges in contact with water. In order to avoid burst effects, it is essential to exceed the critical thickness of the ethyl cellulose film, especially at the edges of the MT2 tablets. For MT1, the critical EC layer thickness was not achieved; thus, no lag time and only a limited sustained release were obtained.
Difference factors distinctly above 15 (Table 8) indicate no similarity in the sodium benzoate release from MT2 10%, MT2 20%, and pellets.
| Parameter | Evaluation | MT2 10%/20% | MT2 10%/EC pellets | MT2 20%/EC pellets |
| Difference factor | “similar” 0–15 | 64 | 56 | 23 |
| Similarity factor | “similar” 50–100 | 57 | 58 | 68 |
Table 8: difference factor and similarity factor for comparison of release from MT2 10%, MT2 20%, and EC pellets.
The different thickness and the condition of the ethylcellulose layers result in significant deviations in the release plots. Otherwise, the similarity factors meet the acceptable range of 50–100, hinting to the weaker evaluation of this parameter. The high coating level of EC pellets (44.4%) with a high specific surface area corresponds more obviously to the higher-coated MT2 20% (similarity factor 68) than to MT2 10% (similarity factor of 58).
3.3.5. Linearization of the release plots for MT2 10% and MT2 20% in comparison with EC pellets
The similarity of the release from the MT2 20% and EC pellets is obvious when comparing the first-order kinetics CoD values of 0.92 and 0.95, respectively (Table 9).
| Coefficient of determination R2 | MT2 10% | MT2 20% | EC pellets |
| Zero-order | 0.47 | 0.73 | 0.90 |
| Square root | 0.75 | 0.85 | 0.88 |
| Sigma Minus plot (first-order) | 0.79 | 0.92 | 0.95 |
| RRSB (first-order) | 0.81 | 0.92 | 0.95 |
Table 9: coefficient of determination of sodium benzoate release from MT2 10%, MT2 20%, and EC pellets with 44.4% ethylcellulose coating.
The lower-coated lot MT2 10% neither refers to zero-order kinetics nor to square root kinetics, but rather to first-order kinetics with CoD values in the range of 0.79 to 0.81. The interaction of the minitablet disintegration with the ethylcellulose swelling, sodium benzoate dissolution, and diffusion out of the pellets presumably leads to first-order kinetics, as in the case of EC pellets with a CoD of 0.95 (Table 9).
4. Conclusions
Drug- and polymer-coated pellets may be compressed into minitablets as well as into normal tablets. Pellets and minitablets compressed into tablets as a multiparticulate drug delivery system offer the advantage of being widely spread in the small intestine for improved drug absorption and increased bioavailability compared to normal tablets. With both tablet types (normal and mini), the sodium benzoate release is delayed depending on the thickness and properties of the ethylcellulose film on the pellets.
Ethylcellulose-coated minitablets obtained through the direct compression of sodium benzoate and excipients are an alternative method for ultiparticulate drug delivery system preparation. The product is characterized by a slow release rate depending on the ethylcellulose film thickness hat may be suitable when prolonged release is required. The variation in release is increased compared to pellets and normal multiple-unit tablets with pellets. Sodium benzoate layering on pellets in the fluidized bed and the subsequent tableting process are feasible for both minitablets and normal tablets, representing an interesting option when a variation in the final pharmaceutical form is required for marketing/patenting reasons.
References
[1] Lennartz, P.; Mielck, J.B. Minitabletting: Improving the compactability of paracetamol powder mixtures. Int. J. Pharm. 1998, 173, 75–85.
[2] Tissen, C.;Woertz, K.; Breitkreutz, J.; Kleinebudde, P. Development of minitablets with 1 mm and 2 mm diameter. Int. J. Pharm. 2011, 416, 164–170.
[3] Hagen, E.; Sandberg Loeding, F.; Mattsson, S.; Tho, I. Use of interactive mixtures to obtain minitablets with high dose homogeneity for paediatric drug delivery. J. Drug Del. Sci. Technol 2016, 34, 51–59.
[4] Rumondor, A.C.F.; Harris, D.; Flanagan, F.; Biyyla, V.; Johnson, M.A.; Zhang, D.; Patel, S. Minitablets: Manufacturing, characterization methods, and future opportunities. Am. Pharm. Rev. 2016, 19, 5–13.
Paper Sections:
Last Submitted Papers:
- Natural coating formulations: IMA is ready for the latest trend.
- New solutions for Nutraceuticals.
- Component washing: better with less.
- Coating conundrum grappling with minitablet film coating myths.
- Influence of material and capsule filling process with Minima on aerosolization performances by DPIs.
- The potential of Croma continuous coater.
- How to enhance tableting production with a paracetamol based formulation.
- The Prexima challenge. Comparison of rotary tablet presses seminar in Leverkusen (Germany), 2-4 July 2019.
- How to perform a good scale-up
- Development of an automated multi-stage continuous reactive crystallization system with inline PATs for high viscosity process
- Prexima 300. Determination of the effect of the pre-compression force on the tablet hardness, obtained at constant value of the main compression force
- FMECA Risk Analysis background for calibrated containment solutions
