
Because many APIs show a limited stability in liquid form, effervescent application has provided a good solution.
Formulation development of Vitamin C based effervescent blend suitable for direct compression.
Introduction
Oral solid dosage (OSD) forms are the most used for medication administration due to their ease of assumption, transport and greater stability; conventionally tablets are more common because, with good formulation, the manufacturing process is simple and with relative low cost. [1]
Active Pharmaceutical Ingredient (API) / Supplements delivered through Oral Dosage Forms may encounter the risk of low absorption. This can be overcome by administering the drug in liquid form. However, because many APIs show a limited stability in liquid form, effervescence application has provided a good solution: drug dissolution and consumption and, at the same time, faster and more complete absorption compared to conventional tablets. Several advantages should be considered: improved patient compliance since there is no need to swallow tablets, less stomach and intestinal irritation, accurate dosing and more reproducible pharmacokinetics are a few examples. Process manufacturing and blend formulation need some specific arrangements in effervescent field:
- Larger tablets
- Complex production and need for specialist packaging materials as well as non-toxic
- Tasteless and water-soluble lubricant
Lubricant is, for this reason, a critical feature that needs to be deeply evaluated: magnesium stearate, as other water-insoluble lubricants, avoids obtaining a clear transparent solution and retards the disintegration and dissolution which is required to be rapid in effervescent tablets. [2] One of the most challenging product development for a formulators is designing a multivitamin formulation which is as stable as possible: several adverse effects can occur due to vitamins stability, which is clearly more difficult in aqueous forms than in solid or oily products. Many formulations showed that there is no significant influent of the auxiliaries (binders, fillers, disintegrants, etc.) on stability of the vitamins themselves if the free water content is limited. The best way to process vitamins, indeed, is direct compression to eliminate water addition, as in granulation: therefore, a proper formulation has to be studied. The combination between excipient and API or supplements in general should guarantee acceptable flowability and compressibility at the same time by ensuring tablet disaggregation.
Aim of the study is the definition of a proper formulation for effervescent vitamin C tablets (ascorbic acid) suitable for direct compression: focus of the analysis, in particular, was the filler choice as it is well known [4] that sugar, fructose, sorbitol or glucose delayed vitamin C hydrolysis (all responsible of instability). Once type of filler and its percentage has been chosen, the target of the experiments was to maximize tablet press output maintaining tablets quality.
Materials and methods
Effervescent ascorbic acid formulations (Table 1) have been well explored thanks to the close cooperation between IMA and Polaris: the target was to obtain an equilibrium between flowability and compressibility.
| Compounds | Formulation (%) | ||||||
| I | II | III | IV | V | VI | VII | |
| Ascorbic Acid | 23.7 | ||||||
| Citric Acid anhydrous | 23.3 | ||||||
| Sodium bicarbonate (Effe-Soda ®) | 11.7 | ||||||
| Aspartame | 0.5 | ||||||
| Dextrose | 19.65 | 0 | 27.51 | 23.58 | 11.79 | 3.93 | 7.89 |
| Functionalised dextrose (EMDEX®) | 19.65 | 39.3 | 11.79 | 15.72 | 27.51 | 35.37 | 31.44 |
| Magnesium Stearate | External lubrication | ||||||
| PEG 6000 | 1.6 | ||||||
| Total | 100 | ||||||
Table 1: Formulations studied.
The most performing formulations have been analysed in terms of technological features: Carr Index, water content (L.O.D.) and particle size distribution have been the characteristics explored. Preliminary trials allowed to define ascorbic acid and effervescent compounds percentages: particular attention was dedicated to functionalised sodium bicarbonate powder to optimize formulation tabletability. Lubricant addition was performed through external lubrication (LUMS, IMA, Italy) to directly insert the minimal quantity of magnesium stearate, as demonstrated, [6] onto punches and dies. Different quantity of dextrose and functionalised glucose (EMDEX®, JRS Pharma) were studied: the best compromise between flowability and compressibility has been chosen for the final formulations. Each blend was obtained through a geometrical blending in tumbler-blender (Cyclops Mini, IMA) by maintaining constant the speed and time: 15 minutes at 15 rpm in detail.
Tableting was performed in rotary tablet press (Prexima 300, IMA) equipped with Euro-D turret hosting 27 stations Eu-D 441 25 mm biconvex round tablets. Feeder was standard with flat paddles installed: the die loading of the blends under analysis was achieved with the standard IMA Prexima tablet press set-up.
First, with all the formulations studied, tableting has been performed at 50 rpm as turret speed with comparable values of pre-compression force (7kN) and main compression force (30 kN). The formulations have been chosen as optimal according to the tablet strength obtained and effervescence behaviour (disintegration time in particular). The selected formulations have been tableted at their best in terms of output, maintaining constant the most critical process parameters: final tablets characteristics and process stability were the results observed to hit the target of the study. Weight and tablet strength stability as well as compression relative standard deviation were the most important parameters to focus on. Parallelly, disaggregation time was always followed to double-match the formulation choice: it has been performed at 37°C until the complete disaggregation of the tablet.
Results
Tablets were always obtained with low standard deviation on weight and with standard tablet press configuration: this means that globally the formulations are suitable for direct compression. During preliminary tests, the use of Effer-Soda (SPI Pharma, Germany) was helpful in developing a formulation for direct compression thanks to the surface modification at sodium bicarbonate particles to enhance stability increasing resistance to humidity and preventing premature effervescence. [5] Adding magnesium stearate (Brenntag, Italy) through external approach and not blended inside the formulation increases the average tablet strength preventing capping defect: LUMS (IMA, Italy) allows to insert the minimum as possible to prevent sticking onto dies and punches, as studied previously. [6]
Technological features measured for Formulation II were 6% and 2.45% as Carr Index and L.O.D. whereas for Formulation VI respectively 7% and 2.84%. Particle size distribution (Figure 2) reflects the different percentage of excipients used.
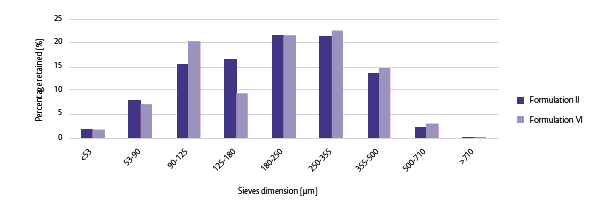 Figure 2: Particle size distribution comparison for Formulation II and VI.
Figure 2: Particle size distribution comparison for Formulation II and VI.
To confirm the choice in terms of filler concentration, tablet strength was analyzed compared to EMDEX percentage for each formulation mentioned (Figure 3).
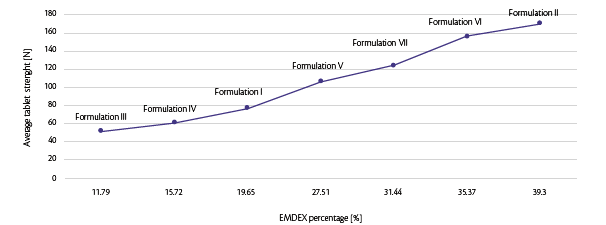 Figure 3: EMDEX percentage vs tablet strength at same turret press speed.
Figure 3: EMDEX percentage vs tablet strength at same turret press speed.
Same turret speed (50 rpm) was first analyzed at same pre-compression and main compression force (respectively 7 and 30 kN): the only variable was the concentration of functionalized glucose (EMDEX) inside the formulation itself.
It is clear that increasing EMDEX percentage, the crushing strength of tablets augments achieving good and defect-free tablets. For low concentration, tablets are pressed with capping tendency and not acceptable hardness, as represented in Figure 3.
It is advisable, in order to reach high machine output, to use at least 90% of EMDEX.
For this reason, only Formulation II and VI were used as reference to study tabletability and effervescence behavior: more in details these two formulations were the focus of the second step of this case study (Table 2).
| PARAMETER | m.u. | VALUE | |||||
| Formulation | II | VI | |||||
| TEST | – | A | B | C | A | B | C |
| Paddle speed | rpm | 35 | 50 | 80 | 35 | 50 | 90 |
| Compression force | kN | 29.9 | 30.3 | 29 | 31.1 | 29.5 | 29.4 |
| Deviation | % | 2.1 | 2.1 | 3.4 | 2 | 1.8 | 2.4 |
| Ejection force | N | 349 | 334 | 419 | 371 | 388 | 474 |
| Turret speed | rpm | 50 | 65 | 80 | 50 | 65 | 70 |
| Production speed | tph | 81,000 | 105,300 | 129,600 | 81,000 | 105,300 | 113,400 |
Table 2: Tableting process parameters at increased output.
The higher use of EMDEX, the higher output can be reached maintaining stable process and defect-free tablets. 113,400 tab/h versus 129,600 tab/h respectively with full-presence of EMDEX and with 90%-10% as EMDEX-Dextrose percentages.
To confirm the hypothesis, tabletability at the turret speed studied was investigated (Figure 4).
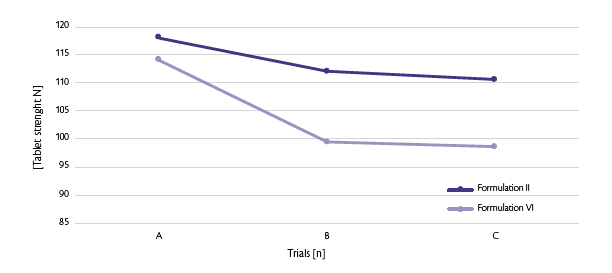 Figure 4: Tablet strength trend.
Figure 4: Tablet strength trend.
All the data are comparable except the test C where the final maximum output was different for the two formulations (80 rpm respectively for Formulation II and 70 rpm for Formulation VI). Disintegration time has been monitored as well for the three tested performed (A, B, C) (Figure 5).
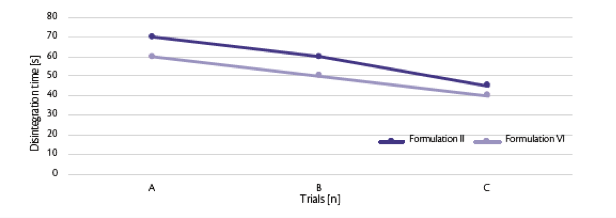 Figure 5: Disintegration trend.
Figure 5: Disintegration trend.
Trial A and B were the performed at same turret press speed: the presence of Dextrose, also in small quantity, decreases the disintegration time. The main reason could be applied to the slightly different crushing strength obtained for the two formulations: higher the tablet strength higher the disintegration time. Even if respected the trend also for Test C, the difference in the time could be due also the turret speed applied: 80 rpm for Formulation II and 70 rpm for Formulation VI. Increased dwell time, clearly obtained at lower turret press speed, reduced the porosity intra-particles and slow-down the disintegration as well.
Conclusions
Effervescent vitamin C formulation has been developed suitable for direct compression: the use of external lubrication approach allowed to increase the tablet strength minimizing defect such as capping. The choice of filler has been crucial: functionalised excipient such as EMDEX improved tablet quality while maintaining an acceptable effervescent behaviour. In the meanwhile, the use of a functionalised sugar permitted to increase tablet press output while ensuring process stability and reliability.
References
[1] Srinath et al., “IJPRD, 2011”, Vol 3 (3), pp. 76-104, May 2011.
[2] Giatti F., https://ima.it/pharma/leading-the-way/
[3] Buhler V., “Vademecum for Vitamins Formulations”, 2nd edition, pp. 75-78.
[4] Buhler V., “Vademecum for Vitamins Formulations”, 2nd edition, pp. 78-79.
[5] https://www.spipharma.com/en/products/functional-excipients/effer-soda/
[6] F. Giatti, S.F. Consoli, F. Bang, T. Cech and C. Funaro, Evaluating the performance of external lubrication to convert the pre-formulated Ibuprofen DC 85 W into a ready-to-use tableting product, “3rd European Conference on Pharmaceutics”, March 25- 26, 2019, Bologna, Italy.
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website


