Thanks to the cooperation between IMA and Labomar, the die-plate adhesion phenomenon has been optimized through different approaches to achieve defect-free tablets when working with a sticky blend.
How to approach die plate adhesion during tableting.
Introduction
Toubleshooting is a form of problem solving, often applied to repair defective products or processes on a machine. It is a logical, systematic search for the source of a problem in order to solve it and make the product or process operational again. Tablet compression is a science and an art, and a host of factors which each contribute to a successful production run. [1]
In pharmaceutical manufacturing, the high level of final product quality is the major driver. Tablets produced should be defect-free and compliant to good manufacturing practice aspects and final-user requirements.
Concerning tableting, the most common defects are classified as visual or functional [2]: visual issues compromise tablet quality without affecting its functionality, something which functional issues affect, such as API availability.
The key to properly managing troubleshooting of the tableting process is to thoroughly investigate the causes of the defects: mostly, it can be approached by exploring the physical-chemical features of the formulation, the selected tableting process parameters or equipment wear even including tablet press parts such as punches and dies.
In the majority of cases, formulation compounds represent a fixed starting point from which any changes can be implemented, whereas tableting process parameters as well as machine condition, can be managed and optimized to their best, combining the knowledge of the machine manufacturer with the experience of the pharmaceutical industry. Stickiness is one of the crucial issues to be faced: it generally happens because, during tableting, powder may stick to the part in contact with the product.
Generally, stickiness occurs on punches and dies since they are directly concerned by the tableting process; therefore die-plate adhesion occurs in particular conditions, mainly related to product features.
An excessively high amount of fine particles in the formula, incorrect environment control or compounds with low melting points (e.g. sugar derivates) are examples of possible causes that generate stickiness on the die plate: the inner heat generation during the process (force application and friction below scrapers and feeding system) accelerates adhesion in the case of a product with the just-mentioned properties. [3]
In this particular case, the standard approach used to solve sticking on punches and dies is palliative: providing a higher quantity of lubricant, introducing external lubrication [4] or specific punch-tip coatings do not improve the final results.
Thanks to the cooperation between IMA and Labomar, the die-plate adhesion phenomenon has been optimized through different approaches to achieve defect-free tablets when working with a sticky blend. In the end, production was optimized also in terms of output.
Materials and methods
For the purpose of this study, a sticky formulation of a chewable tablet has been optimized; the latter includes a probiotic and a prebiotic (oligosaccharides – OS) as active ingredients, with the function of promoting the balance of the intestinal flora, and polyols as fillers, due to their good flowability, their high solubility and their sweet taste.
In detail, prebiotics are defined as a “non-viable food component that confers a health benefit on the host associated with modulation of the microflora”. [5] OS are dietary fibres, i.e. carbohydrate polymers with a ≥3 degree of polymerisation, that are not digested or adsorbed in the intestine; rather they are fermented by the gut microbiota producing energy for their survival. [6] OS present in the formulation is a fine powder that is highly hygroscopic and presents a low melting point because of its sugary nature. Polyols, also defined as polyalcohols or sugar alcohols, are typically derived from hydrogenation of sugars (mono-, di- or polysaccharides) [7], for this reason they generally appear to be moderately to highly hygroscopic powders. Polyols present in this formulation are characterised by a melting point ranging between 90 and 110°C.
Table 1 summarizes the qualitative composition of the formulation, while Table 2 shows the tests and related methods performed on the mixture.
| Active Ingredient (A) / Excipient (E) | Description | Percentage (%) |
| A | Probiotic | <5 |
| A | Probiotic | >25 |
| E | Polyols | >60 |
| E | Lubricant | > 0.5 |
| E | Flavours | as required |
Table 1: Qualitative and quantitative composition of the mixture.
| Test | m.u. | Method |
| Particle Size Distribution (PSD) | µm | Laser diffraction particle size analyser Analysette 22 equipped with a Ø 1.5 mm funnel (Fritsch GmbH MicroTec plus / NanoTec plus, Idar-Oberstein, Germany). |
| Water activity | – | Lab touch – aW SYSTEM (NOVASINA, Lachen, Switzerland) |
| Loss On Drying | % | 5 grams, 80°C, 10 minutes (i-THERMO 163M, 64M) |
| Density | g/ml | Italian Pharmacopoeia XII ed |
| Carr Index (CI) | % | Italian Pharmacopoeia XII ed |
Table 2 : Test and related methods performed.
A Prexima 300 rotary tablet press was used for process optimisation. The machine was equipped with a Euro-D turret hosting 27 stations with round 14.5 mm punches. The feeder installed had two paddles inside with a flat geometry and a loading cam of 12 mm was selected.
The distance between feeder and die plate was 0.05 mm, as a good starting point.
In this condition, stickiness on the die plate was evident (Figure 1) generating defects on the tablets themselves: in detail, a darker spot appeared on the surface due to the residual powder that was recovered by the machine and compressed again.

Figure 1: Die plate adhesion.
To manage this issue, approaches were made at different levels (Table 3):
1. Loading cam;
2. Feeder geometry;
3. Distance between die plate and feeder
| Tableting process parameters | m.u. | Values | |||
| Loading cam | mm | 8 | 12 | ||
| Feeder geometry | – | Flat-Flat | Flat-Round | ||
| Distance Feeder-Die plate | mm | 0.05 | 0.10 | 0.12 | 0.15 |
Table 3: Tableting process parameters investigated during
To evaluate the efficiency of the adjusted parameters, the most important tableting process responses (main compression force and its relative standard deviation, output reachable without defect, time of defect occurrence) as well as the tablets’ visual aspect were evaluated (Table 4).
| Parameters | m.u. | Value | |||||
| Test | – | 1 | 2 | 3 | 4 | 5 | |
| Paddle profile | – | Flat-Flat | Flat-Round | Flat-Round | Flat-Round | Flat-Round | |
| Loading cam | mm | 12 | 8 | 8 | 8 | 8 | |
| Main compression force | kN | 23 | 22.5 | 22.6 | 24.2 | 24.4 | |
| CF standard deviation | % | 7 | 4.3 | 4 | 3.8 | 3.7 | |
| Turret speed | rpm | 30 | 15 | 20 | 25 | 25 | |
| Production output | tph | 48,600 | 24,300 | 32,400 | 40,500 | 40,500 | |
| Distance feeder-die plate | mm | 0.05 | 0.05 | 0.1 | 0.12 | 0.15 | |
| Good tablet appearance | (Y/N) | N | N | N | N | Y | |
| Time of defect occurrence | Y/N (min) | Y,2 | Y,10 | Y,30 | Y,60 | N | |
Table 4: Tableting process responses and tablet appearance.
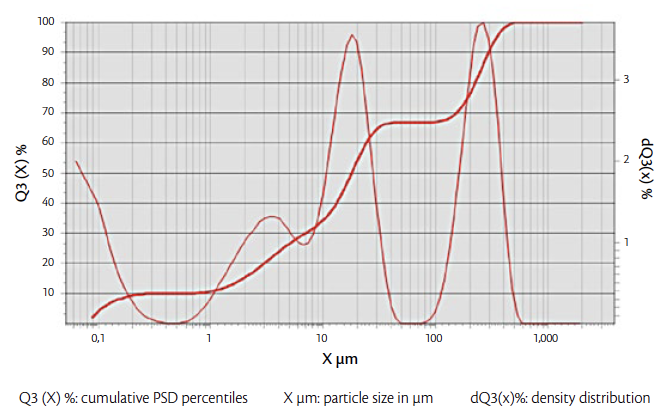
Figure 2: PSD graph of the mixture and its cumulative oversize curve.
Results and discussion
In order to examine the die-plate adhesion phenomenon, tests described in Table 2 were performed to evaluate the technological properties of the mixture.
Results of these analyses are included in Table 5.
| Test | m.u. | Results |
|
Particle Size Distribution (PSD) |
µm | D10= 0.7 D50= 17.8 D90=295 (*Figure 2) |
| Water activity | – | 0.223 |
| Loss On Drying | % | 0.78 |
| Bulk density | g/ml | 0.67 |
| Carr Index (CI) | % | 13 |
Table 5: Results of powder characterization tests.
Results of investigation led to assume that formulation compounds caused the stickiness on the die plate. In particular, oligosaccharides (OS) tended to melt if they underwent excessive friction (with heat generation as a consequence) inside the feeder and between feeding system and die plate.
The approaches that were explored allowed for gentler handling of this compound producing, as a consequence, less friction and heat generation. Having a round profile on the paddle at the right position inside the feeder smoothly scraped the extra powder dosed inside the dies, whereas a smaller loading cam reduced the powder dosed back inside feeder. The combination of these two parameters reduced powder movement inside the feeder, a factor that could concur in heat generation.
The most important process parameter managed was the distance between the feeder and the die plate: increasing this value in order to be comparable with the particle size distribution of the blend allowed for a gentle passage of the powder below the feeder without generating friction and heat. The result is defect-free tablets and a perfectly clean die plate without any adhesion.
Conclusions
Troubleshooting in die-plate adhesion is generally challenging because the main reason is directly related to formulation characteristics.
Gentler blend management was the lynchpin to reduce the possibility of generating friction and heat: round paddle geometry, reduced refilling inside the die feeder and distance accurately studied according to particle size distribution allowed for process optimisation that stabilised the overall tablet press efficiency and quality of the tablets.
References
[1] Kirsch D., Fixing Tableting Problems, “Pharmaceutical Technology”, 39 (5), 2015.
[2] “Tablet Resolution Handbook“, 2014.
[3] Adhnikart B., Bhandanrt B., Howers T., Truong V., Stickiness in Foods: A Review of Mechanisms and Test Methods, “International Journal of Food Properties”, 4(1), 1- 33, 2001.
[4] Giatti F. et al., External lubrication application on a double-side rotary tablet press: how to set up the equipment and minimise time losses, https://ima.it/pharma/white-papers/?cat=24
[5] “FAO Technical Meeting on Prebiotics”, Rome, September 2007.
[6] Davani-Davari, Negahdaripuor, et al, Prebiotics: Definition, Types, Sources, Mechanism, and Clinical Applications, “National Library of Medicine”, MDPI, 2019.
[7] Małgorzata G., Sugar Alcohols, “Encyclopedia of Food Chemistry”, pp. 265-275, 2019.

In collaboration with:

Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website