
The Extrusion-Molding-Coating (EMC) machine is equipped with spectroscopic-based PATs for the in-line monitoring of drug product quality.
Continuous Manufacturing of solid oral dosage forms by integration of Raman spectroscopy to an Extrusion-Molding-Coating (EMC) unit
Introduction
Process Analytical Technologies (PATs) are intended to support innovation and efficiency in pharmaceutical development, manufacturing, and quality assurance. The framework is founded on process understanding to facilitate innovation and risk-based regulatory decisions by both industry and regulatory agencies.
Lately, regulatory agencies have been encouraging manufacturers to use a framework of PATs to develop and implement effective and efficient innovative approaches for pharmaceutical manufacturing.
The purpose of using PATs is to design and develop dynamic manufacturing processes that can compensate for variability in both raw materials and equipment to consistently ensure a predefined quality at the end of the process. The objective of the PAT application is to monitor and control the product quality in real-time. Thus, downstream and upstream operations can be adjusted accordingly without delay. PATs aim to ensure that critical sources of variability affecting a process are identified, explained and managed. Therefore, a real-time release testing (RTRT) strategy should be based on a firm understanding of the process and of the relationship between process parameters, in-process material attributes and product attributes. Then, closed-loop control allows fine-tuning of process parameters based upon thorough knowledge of how the components and related processes affect the final product. The PAT ideology is in accordance with the fundamental principle that quality should not be tested, but rather be built into the product by design. The association of PATs, sensors and an integrated control system enable the design and development of dynamic manufacturing processes. Indeed, all variabilities affecting the process might be identified, explained and managed, to ensure a high and consistent product quality overtime by automated and real-time measurements. [1–3]
Process context
The Extrusion-Molding-Coating (EMC) machine used for continuously manufacturing molded coated tablets is equipped with spectroscopic-based PATs for the in-line monitoring of drug product quality attributes by non-destructive and automated methods.
The drug substance and a pre-blended mixture of excipients are continuously fed into the EMC through two loss-in-weight feeders yielding to a formulation composed of 70 wt% drug load and 30 wt% of excipients (not disclosed here for a client confidentiality agreement). For this study, as the focus is on tablet core quality monitoring, only the EMC extrusion and molding unit were operated without the coating unit.
The PAT used for this case study is a non-contact Raman probe (Kaiser®, PhAT probe) placed in the transfer manifold section of the EMC, which integrates the twin screw extruder with the injection molding unit (Figure 1).

Figure 1: Schematic view of the Extrusion-Molding-Coating (EMC) machine equipped with PATs.
Raman spectroscopy is used in this location to confirm the identity of the Active Pharmaceutical Ingredient (API) along with its crystallinity and crystal form. This method has a few steps that need to be set during development and integration. For instance, the sample interface must be exempt from any other light sources, including in situ fluorescence that will interfere with measurements. In addition, the non-contact Raman spectroscopy procedure is carried out on hot and moving material, so the concentration of analyte must be sufficiently high. Finally, to obtain constant and reliable measurements, the sample interface must be kept consistent (pathway length of Raman light, angle between the light beam and matter). Nevertheless, the use of the proposed Raman analytical procedure for drug product is non-destructive, and it provides continuous measurements of the final dosage form without requiring sampling from the process stream. In addition, its integration with the control system can engage automated corrective action in the case of process deviation and as part of a real-time release testing strategy.
A two-phase approach was developed to achieve the final goal of the analytical procedure, which is to confirm the identity and crystallinity of the drug product in-line and real-time. The first phase is exclusively composed of off-line measurements by two primary methods (DSC, XRPD) and a secondary method (Raman).
This phase is used to:
• confirm the crystalline state of the API in the formulation by different analytical techniques,
• verify the conformity or non-conformity of samples by Raman off-line.
The second phase compares the off-line Raman spectra (obtained during phase one) with the in-line spectra to assess the equivalency of these analytical procedures and verify the reliability of the Raman spectroscopy procedure.
Off-line characterization of API and excipients
The analysis of the same sample by different primary analytical techniques (XRPD and DSC) versus Raman allows the verification of the conformity or non-conformity of samples by Raman off-line. Thus, the API and the excipients were analyzed individually by all the three techniques.
Differential Scanning Calorimetry (DSC) was used for assessing the composition and purity of mixtures, as well as evaluating the crystal form. Figure 2 (a) shows the heat flow of the formulation components as individual materials and Figure 2 (b) presents the heat flow of the physical mixture formulation (powders blend before being processed by the EMC) and the final drug product (i.e., ground tablets after being extruded and molded).
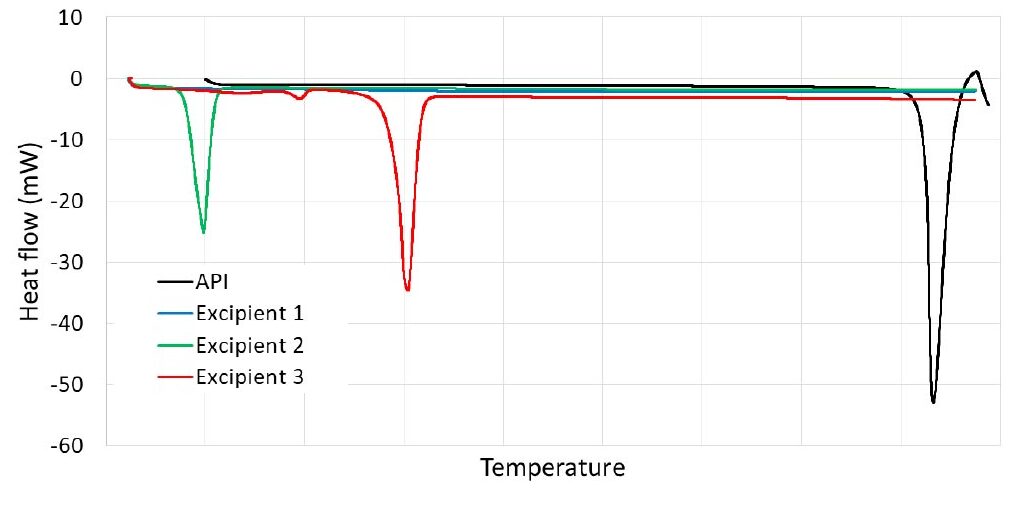 Figure 2: DSC analysis of (a) API and excipients; (b) API, the physical mixture of the formulation, and the drug product.
Figure 2: DSC analysis of (a) API and excipients; (b) API, the physical mixture of the formulation, and the drug product.
The DSC profile of the API did not show any major heat capacity change but a melting peak, demonstrating API crystallinity. Conversely, the DSC profile of the main polymeric carrier did not show a sharp peak due to its amorphous nature. A melting peak was observed for the other two crystalline excipients. The same formulation from both pre-extrusion-molding (physical mixture) and post-extrusion-molding (ground tablets) were analyzed by DSC (Fig. 2 (b)). Due to the materials mixing into a uniform physical mixture, the phase transition of the API was shifted toward lower temperatures but still showed a defined peak of crystalline melting behavior. The drug product (ground tablets) showed a profile almost overlapping to the physical mixture, showing that the API preserves its crystalline structure even undergoing the extrusion-molding process.
X-ray powder diffraction (XRPD) experiments were performed on the same samples:
• individual components of the formulation,
• physical mixture
• the drug product.
XRPD analysis of individual components resulted in distinct fingerprints for the API and the two excipients (not shown for confidentiality reasons); and a typical amorphous halo was obtained for the main polymer matrix. The physical mixture and the drug product were analyzed, and a few observations were made. First, the relative intensities between the specimen and the reference API may differ due to preferred orientation effects in the sample. Second, the agreement of the 2θ-diffraction angles between the formulation, drug product, and reference API is within 0.2°, which means that they share the same crystal form according to USP 41-N F36 Characterization of Crystalline and Partially Crystalline Solids by X-ray Powder Diffraction (XRPD) <941>. In conclusion, the diffractograms of both physical mixture and ground tablets superimposed align almost perfectly with the pure API diffractogram. In conclusion, the API in the drug product was found to be crystalline and of the same crystal form as the starting drug substance.
In addition to DSC and XRPD analyses, complementary off-line Raman spectra were collected for individual components of the formulation, the physical mixture, and the drug product.
The spectrum of the API displayed a maximum of absorption at a specific wave number with a high specificity for that crystal form. The main polymeric carrier shows a high Raman signal spread out without any sharp peaks, which is typically obtained from amorphous or semi-crystalline polymers (as previously confirmed by DSC and XRPD). The other excipients, characterized by crystalline nature, showed intense, diagnostic peaks. These results are in agreement with those obtained from DSC and XRPD.
The formulation physical mixture and the drug product samples were analyzed off-line by Raman and compared to the pure drug substance. The overall fingerprint regions obtained by Raman for the formulation physical mixture, drug product, and drug substance were almost overlapping, except for the constant greater (and expected) intensity observed with the pure drug substance, as shown in Figure 3. The maximum intensity of these three samples was found at the same specific wavenumber being consistent with previous results. In conclusion, the API in the drug product was identified to be crystalline by Raman and of the same crystal form as the drug substance.
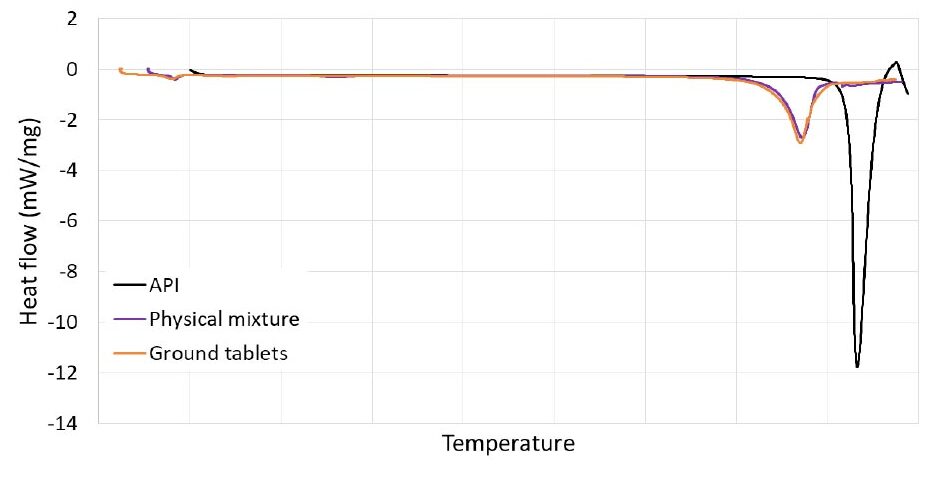
Figure 3: Raman analysis of API, formulation physical mixture, and drug product. The figure shows relative Raman intensities as a function of the Raman shift in wavenumbers (cm-1) (relative scale) after a Pearson’s baseline correction.
In-line Raman analysis
The Raman was integrated with the EMC unit to monitor in-line the crystallinity of the API in the product. The API and excipient powders are fed and mixed in the twin-screw extruder, where they create a homogeneous fluid polymeric melt and suspension of crystalline API thanks to heat and shear. This fluid is conveyed through the manifold toward the injection molding section to produce tablets. The Raman probe was placed in the transfer manifold which has a sealed optical-grade window for non-contact spectroscopic analysis (refer to Figure 1). The probe is an independent component and could be plugged/unplugged without interfering with process operation/stream.
The Raman spectra of the API was obtained in-line and off-line from equivalent material and are compared. Thus, two different materials were analyzed with:
• a flowing hot-melt extrudate for in-line analysis and
• a ground powder of molded tablets at room temperature for off-line analysis (Raman, and also DSC and XRPD).
The tablets were collected according to the mean residence time determined by Residence Time Distribution (RTD) studies previously performed to correspond to the same hot-melt extrudate monitored in the transfer manifold. Their Raman spectra are compared in Figure 4 to confirm the crystallinity of the API in-line and the equivalence of the two Raman approaches.
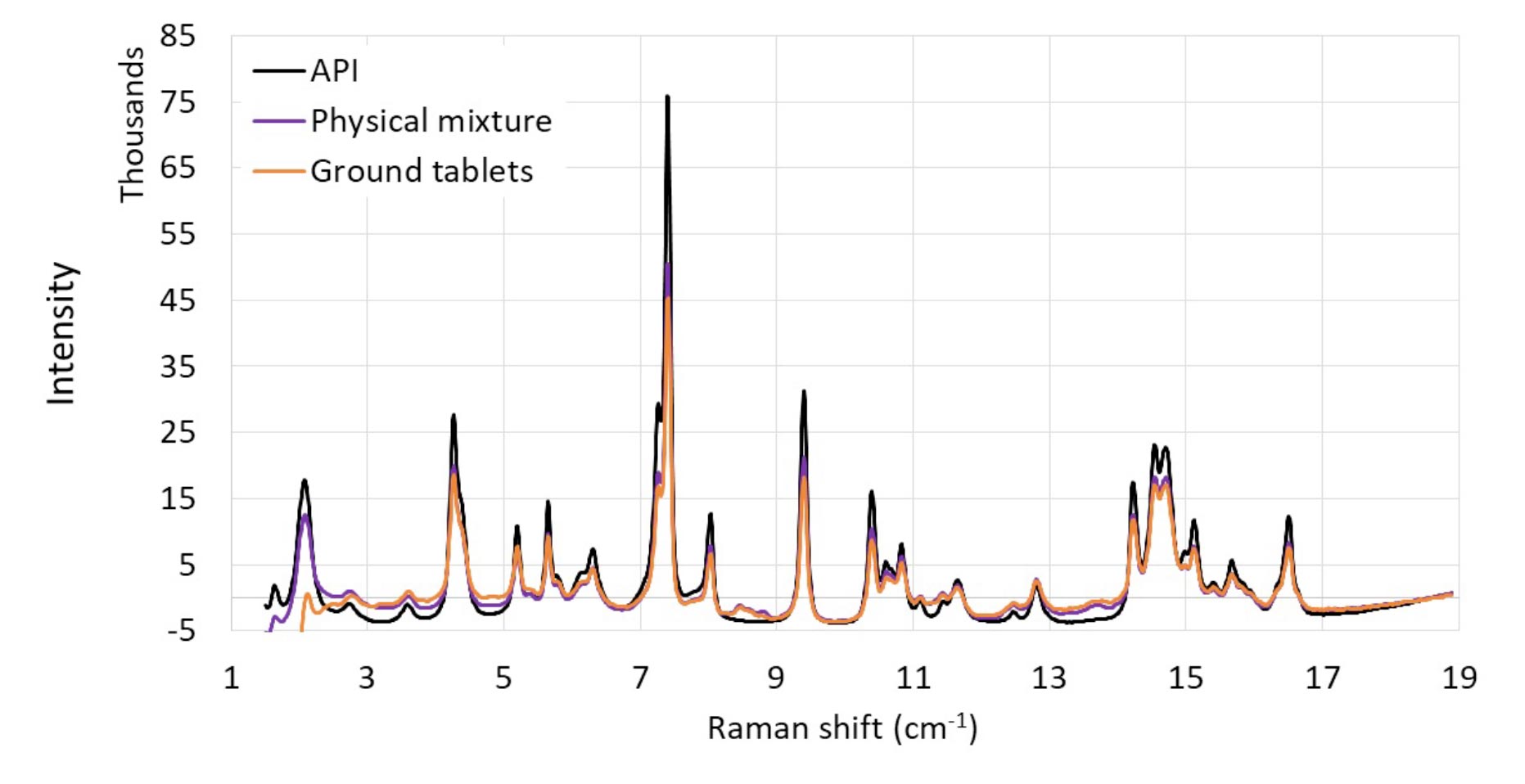
Figure 4 Raman spectra of the drug product in-line and off-line. In-line testing of the drug substance is shown in red. Off-line testing of the drug substance is shown in blue. Raman shift in relative scale.
Their Raman spectra are compared in Figure 4 to confirm the crystallinity of the API in-line and the equivalence of the two Raman approaches.
Distinct Raman spectra were obtained (Figure 4), and similar features were shared between the in-line and off-line analyses of the drug product. Both spectra contain major API peaks at the same wavenumbers, which confirms the crystallinity of the drug (in agreement with off-line analyses by DSC and XRPD). In addition, this study confirms the feasibility of monitoring the crystallinity of the drug product in-line by Raman spectroscopy in the EMC unit.
In-line continuous measurements of the drug product crystal form by Raman were performed with some intentional process deviations. During the test, a constant throughput of one kilogram per hour was used, alternating the feeding of a placebo formulation and the API formulation. The peak height of the most intense spectral peaks for the API were monitored over time.
Referring to the graph shown in Figure 5, at time 0 (t0) the placebo formulation was fed, and no Raman peaks were detected. At 0.7 hours, the formulation was fed to the EMC system leading to a sharp increase of the API signal immediately. Subsequently, at time 1.6 hours the placebo formulation was fed again, leading to a steep decrease of the API signal. This was followed by a repetition of feeding the formulation and then the placebo mixture at time 1.9 hours and 2.7 hours, respectively, leading to similar results for the three major Raman signals of the API.
Figure 5 In-line continuous Raman monitoring of the process. The feeding formulation contains crystalline API and the purging material is a placebo formulation.
Conclusion
The analytical data reported demonstrate that the developed Raman analytical procedure can be implemented in-line to continuously monitor the crystallinity of the drug API in the hot-melt extrudate. The specificity of the Raman procedure was validated with samples determined to be conforming off-line (i.e., physical mixture, ground tablets) together with samples determined to be non-conforming in-line (i.e., placebo formulation) and samples determined to be conforming in-line (hot-melt extrudate of the formulation). Thus, the crystallinity of the drug product was verified by Raman spectroscopy off-line and in-line.
This spectroscopic analytical technology provides quick, real-time measurements that verify the identity of the molecule and its crystal form. In addition, this real-time information is valuable for maintaining the process in a state of control and monitoring the critical quality attributes of the final drug product.
References
[1] Food and Drug Administration (FDA), Guidance for Industry: PAT – A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance,(2004).
[2] European Medicines Agency, Guideline on Real Time Release Testing, EMA/CHMP/QWP/811210/2009-Rev1, Comm. Med. Prod. Hum., Use 44, pp. 1–10, 2012. [3] Jiang, M. et al., Opportunities and challenges of real-time release testing in biopharmaceutical manufacturing, “Biotechnol. Bioeng”, 114, pp. 2445–2456, 2017. [4] IMA & Continuus Pharmaceuticals, Feasibility studies of Continuous Manufacturing of Injection Molding Tablets via Extrusion-Molding-Coating (EMC), “Pharm. Technol. US”, 2021. [5] Hu, C. et al., ., ed modular assembly line for drugs in a miniaturized plant, “Chem. Comm.”, 56, pp. 1026–1029, 2020. [6] Testa, C. J. et al., Design and Commercialization of an End-to-End Continuous Pharmaceutical Production Process: A Pilot Plant Case Study, “Organic Process Research & Development”, 24(12), pp. 2874–2889, 2020.
More IMA Active Case Studies
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website


