
The Prexima challenge @ APV Expert Workshop Tableting
Three years after its official market launch, Prexima 300 wins the challenge with the major tablet press manufacturers at APV Expert Workshop Tableting.
The Prexima challenge @ APV Expert Workshop Tableting
Three years ago, we made a bet. We bet that we could compress all our expertise and design capability to create a tablet press just as good as the ones of the major tablet press manufacturers. Or even better. Three years after the official launch of Prexima 300, numbers based on a third party practical demonstration show that we won the bet.
The APV Workshop formula: 5 tablet presses, 3 formulations, 2 compression studies
The workshop was organized by APV – International Association for Pharmaceutical Technology at the Technical University of Dortmund with the aim to have a direct comparison between different tablet press performance. All major tablet press manufacturers were called to participate in the challenge. Those who accepted it were Bosch, Fette, Kilian, Korsch. And IMA Active with Prexima 300.
APV supplied three formulations having different characteristics: plastic, elastic and brittle. With all three formulations two different compression studies were to be carried out:
- A compaction study, reproducing the R&D activity of product development and aimed at measuring tablets characteristics when compressed at a given speed but with different compression forces.
- A stability study, aimed at determining the best performance of the tablet presses in production conditions.
| Compaction study | Stability study | |
| Tablet weight | 250 mg | 250 mg |
| Compression force | 5,10,20 and 40 kN | Set by supplier, to product a 100 N tablet hardness |
| Output | 100,000 tablet/hour | As much as possible |
| Samples | 50 tablets | 50 tablets |
| Sampling criteria | After 15 minutes for each compression force | every 25,000 manufactored tablets |
Stability study: the outcome
Fifty tablets were sampled every 25,000 and weight and tensile strength were measured. The relative standard deviation of the mean values of weight made immediately evident that the performance of our Prexima 300 was the best with all product formulations.

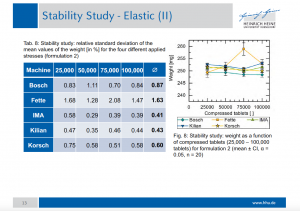
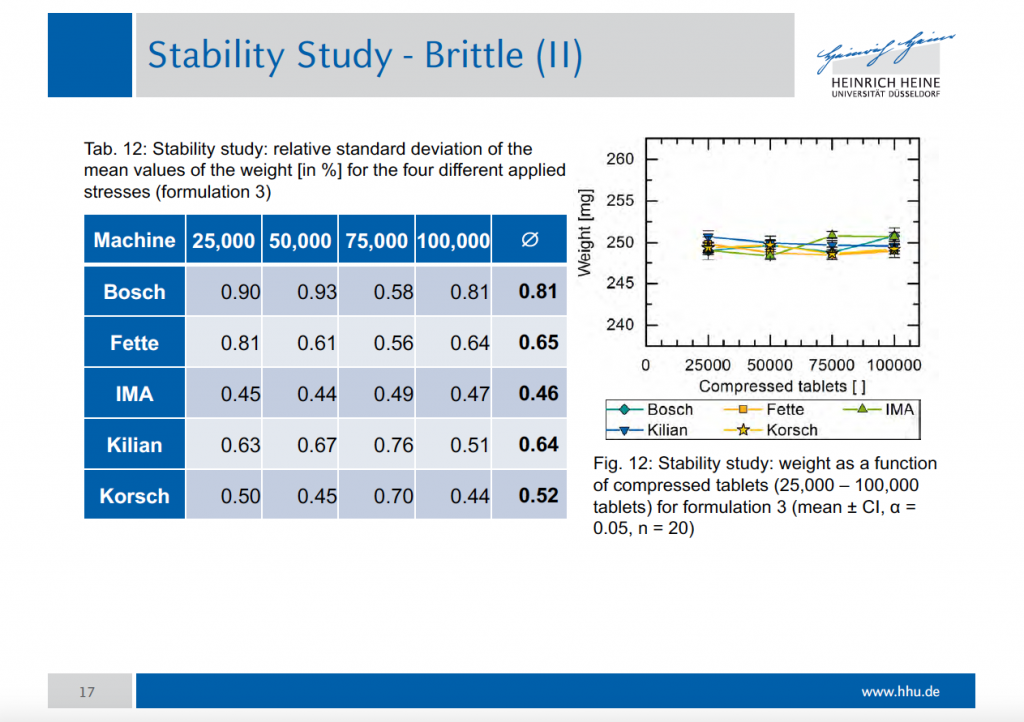
The results of the stability study prove that Prexima 300 performs the best stability of tablets weight in production conditions and with different formulations.
Compaction study: the outcome
Considering the results of the compaction study, the same outcome is obtained: Prexima 300 achieves excellent performance in terms of speed/quality of tablets.
Conclusions
The tests carried out with APV formulations have demonstrated that Prexima 300, just three years after its market launch, is capable of obtaining the same performance – if not superior – to those of the major competitors. The stability study particularly highlights the machine sturdy structure: its pre- and main compression rollers, fixed on both sides inside the triangle of rigidity and having the same diameter of 250 mm, allow manufacturing high quality tablets, without affecting machine speed.
Other updates
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website


