Quick access to IMA Sectors:
Pharma | Food & Dairy | Confectionery | Tea & Beverage | Coffee | Personal & Home Care | Tissue & Nonwoven | Automation | Tobacco | E-Commerce |Quality by design cycle optimisation and equipment capability determination for vaccine placebo.
Introduction
This case study outlines the QbD-based optimisation of a legacy lyophilisation cycle for a vaccine placebo during tech transfer to a new IMA LyoMax 40 unit. The goals were to reduce cycle time, eliminate microcollapse defects, and confirm equipment suitability. Using pilot-scale studies, a design space was developed to guide cycle improvements, followed by equipment characterisation to ensure the new lyophiliser could sustain the optimised process.

Challenge Outline
Customer produced a lyophilised vaccine with a legacy cycle using an older, non-IMA lyophiliser. On purchasing a new, commercial-scale lMA lyophiliser (LyoMax 40, 40m2), Customer required a tech transfer of the cycle to maintain product critical quality attributes (CQAs). A quality by design (QbD) approach was selected to facilitate cycle optimisation efforts. Initial studies showed the occurrence of microcollapse in the lyophilised vaccine product which needed to be addressed. The newly purchased IMA Lyomax 40 would then need to be characterised to determine if changes were required to the new, optimised cycle.

Can the cycle be optimised?
Can the defect be removed?
Can the lyophiliser support the modified cycle parameters?
Objectives

| Expected outcomes |
| A new, optimised cycle which provides equivalent CQAs while reducing defect occurence that can be sustained in the new lyophiliser. |
| Approach |
|
Use IMA’s pilot-scale lyophiliser, the LyoFast 3 (2.3 m2), to conduct a baseline cycle using Customer’s legacy cycle With data collected from baseline cycle, create a Design Space to facilitate the QbD Cycle Optimisation [For more details on design spaces and QbD Cycle Development, check our previous posts!] Make changes to the legacy cycle, assisted by the design space, to shorten cycle time and avoid defect occurrence thus creating a new, optimised cycle Execute a minimum controllable chamber pressure (MCP) test in the new lyophiliser to characterise it [For more details on Equipment Characterisation, check our previous posts!] |
Objectives 1 & 2: Reduce length of legacy cycle & occurrence of microcollapse defect
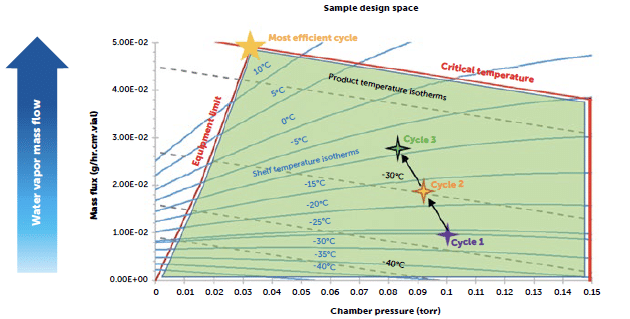
After conducting baseline studies using the legacy cycle, a design space was created for QbD cycle optimisation. Subsequent cycles were executed using modified parameters across all freeze drying steps. As a result of changes made in the drying steps, cycle time was reduced by 20%. Changes made to the freezing step led to elimination of defect occurrence.
Overall cycle time reduced by 20% using design-space-assisted modifications to drying steps.

Objective 3: Perform equipment characterisation test
By performing a minimum controllable chamber pressure (MCP) test, it was determined that the new lyophiliser was able to sustain low enough pressures and have a suitable equipment limit to sustain the newly optimised cycle parameters.
 |
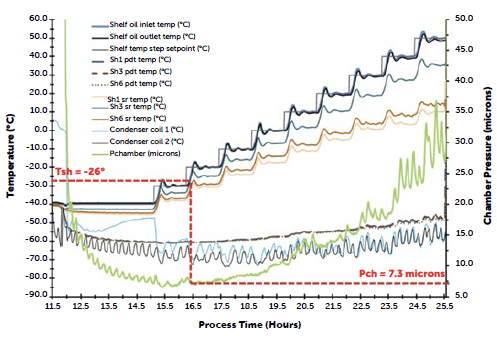 |
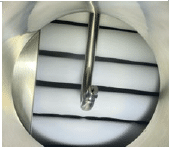
| Loaded Lyomax 40 with thermocouples installed | Sample MCP cycle data & ice accretion on condenser coils at end of test |
 |
Summary
By applying QbD cycle development principles, the Customer’s legacy vaccine cycle was modified to create a new, optimised cycle with lower overall cycle time and no microcollapse defect occurence. On testing the Customer’s newly purchased lyophiliser, it was determined to be sufficiently suited to execute the new cycle parameters, both in terms of controllable chamber pressure and sublimation rates.
You can access further literature at lab4life.ima-pharma.com