Quick access to IMA Sectors:
Pharma | Food & Dairy | Confectionery | Tea & Beverage | Coffee | Personal & Home Care | Tissue & Nonwoven | Automation | Tobacco | E-Commerce |LYOMAX
Freeze-drying involves the removal of solvent such that the molecular structure of the drug’s active ingredient is least disturbed, thus providing a dried drug product that is stable over time.
Freeze-drying involves the removal of solvent such that the molecular structure of the drug’s active ingredient is least disturbed, thus providing a dried drug product that is stable over time.
Beyond Limits
Although freeze-drying is one of the oldest techniques in the pharmaceutical industry for the preservation of unstable materials, there has been an increasing demand for lyophilized products and equipment, in particular in the last decade. In fact, today, about half of all sterile injectables are lyophilized.
Today’s freeze-drying process demands increased process control, with reduced processing time augmented with upgrades in equipment to remain compliant with a changing regulatory landscape. To accommodate such growth, IMA Life uses its in-depth process knowledge to make freeze-drying faster, safer, cheaper, and compatible with all industry requirements and challenges.
LYOMAX configurable options and custom designs provide the optimal solution for each application.
LYOMAX meets the highest possible technical requirements, the strictest standards of the global licensing authorities, and complies with all the current guidelines such as GAMP, cGMP, FDA etc.
Sustainability

IMA LYOMAX freeze dryers embody the requirements of the pharmaceutical industry for faster, safer, cheaper and more ecologically friendly processes:
• The IMA Life patented FUSION™ shelves reduce energy consumption typically in heating, cooling and steam sterilization thus achieving a reduction in energy costs and fast temperature response. The FUSION™ shelves allow for a 20% reduction in weight leading to reduced energy consumption during freeze drying and sterilization.
• PAT tools reduce overall process time and energy consumption.
• IMA Life is developing innovative eco-friendly refrigeration for freeze dryers, KRYOAIR. KRYOAIR is a new 100% green refrigeration technology using the ultimate natural refrigerant “air”. The system is based on a series of compression/cooling/expansion steps of air, which can be cooled to a temperature as low as -100° C.

LYOMAX Highlights
IMA Life's FUSION™ - FUSION PLUS™ shelves
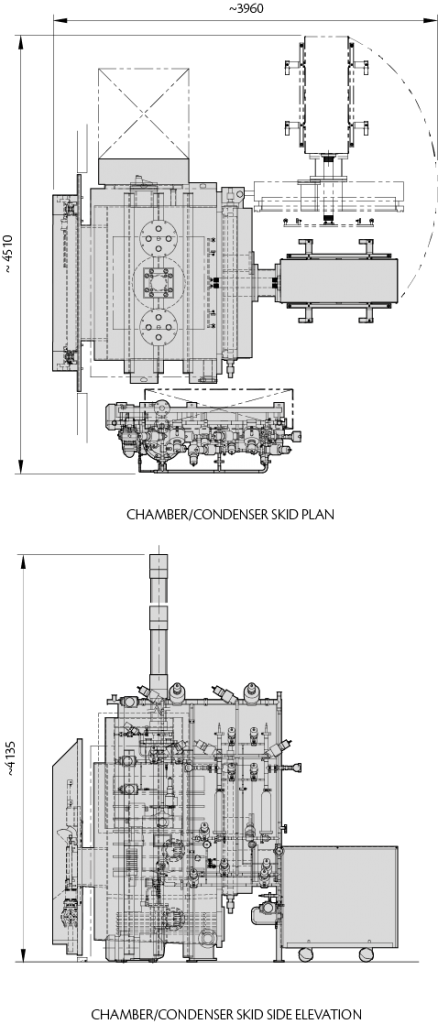
- Technical Data
- Machine Layout
| Model | Number of shelves | Vial pack area Manual loading (m2) |
Vial pack (wxd) Manual loading (mm) |
Vial pack area Auto loading (m2) |
Vial pack (wxd) Auto loading (mm) |
Condenser capacity (kg @12.7 mm ice) | Compressor configuration | LN2 configuration | KryoAir configuration |
| LYOMAX 1 | 5 | 1.4 | 455 x 615 | NA | NA | 28 | 2 x 5Hp Bitzer | LF25/12 | 1 x 23T-0 |
| LYOMAX 3 | 5 | 2.8 | 615 x 916 | 2.6 | 615 x 837 | 70 | 2 x 8Hp Bitzer | LF25/12 | 1 x 23T-0 |
| LYOMAX 5 | 6 | 5 | 916 x 916 | 4.6 | 916 x 837 | 97 | 2 x 12Hp Bitzer | LF25/12 | 1 x 23T-1 |
| LYOMAX 7 | 7 | 7.8 | 916 x 1,220 | 7,.3 | 916 x 1,141 | 143 | 2 x 16 Hp Bitzer | LF50/20 | 1 x 23T-1 |
| LYOMAX 10 | 9 | 10 | 916 x 1,220 | 9.4 | 916 x 1,141 | 194 | 2 x 20Hp Bitzer | LF50/20 | 1 x 23T-1 |
| LYOMAX 15 | 10 | 14.5 | 1,220 x 1,220 | 13.9 | 1,220 x 1,141 | 273 | 2 x 30Hp Mycom | LF50/20 | 2 x 23T-1 |
| LYOMAX 20 | 11 | 20.5 | 1,220 x 1,524 | 19.4 | 1,220 x 1445 | 375 | 2 x 30Hp Mycom | LF50/20 | 1 x 80T-0 |
| LYOMAX 25 | 14 | 26 | 1,220 x 1,524 | 24.6 | 1,220 x 1,445 | 476 | 2 x 50Hp Mycom | LF100/50 | 1 x 80T-1 |
| LYOMAX 30 | 13 | 30.2 | 1,524 x 1,524 | 28.6 | 1,524 x 1,445 | 552 | 2 x 50Hp Mycom | LF100/50 | 1 x 80T-1 |
| LYOMAX 35 | 13 | 36.3 | 1,524 x 1,830 | 34.7 | 1,524 x 1,751 | 635 | 2 x 50Hp Mycom | LF100/50 | 1 x 80T-1 |
| LYOMAX 40 | 15 | 41.8 | 1,524 x 1,830 | 40 | 1,524 x 1,751 | 731 | 2 x 50Hp Mycom | LF100/50 | 2 x 80T-0 |
| Minimum shelf temperature (5cst/1/6cst oil) | -55C/-60C | -65C | -60C | ||||||
| Minimum condenser temperature | -75C | -100C | -75C | ||||||