
Based on the successful LYOMAX industrial FD concept, MINIMAX is a GMP, small-scale freeze dryer developed to address the demands involved in handling small production batches.
Based on the successful LYOMAX industrial FD concept, MINIMAX is a GMP, small-scale freeze dryer developed to address the demands involved in handling small production batches.
Maximise your opportunities
The MINIMAX series provides scalable lyophilization technologies from formulation to full commercial production. Process analytical technologies (PAT) also help manufacturers to find desirable conditions and parameters for perfect drying outcomes. Pilot-scale freeze drying is the optimal solution for scale up operation volumes prior to industrial scale production or even for small production batches. The aim is to achieve drug commercialisation objectives and bring parenteral products safely and quickly to market by enhancing product and process knowledge. This ensures superior product quality and uniformity and creates a data-rich environment ensuring successful operation of all freeze-drying system sizes.
MINIMAX benefits from all-round performance, a high level of redundancy and various configurable options making it a reliable and versatile solution for each application. MINIMAX can be integrated with isolation technology and automatic vial-loading systems. The unit is pre-arranged to support the processing of non-aqueous solvents and high potent products.

MINIMAX Highlights
IMA Life's FUSION™ - FUSION PLUS™ shelves
- Technical Data
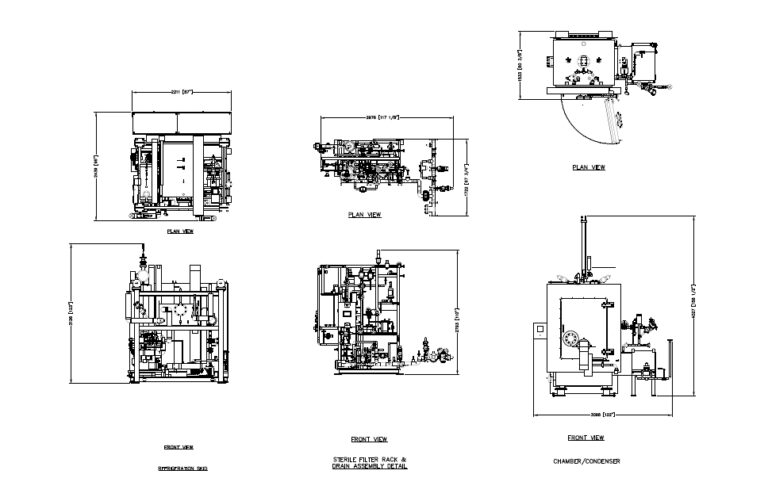
- Machine Layout
| Model | Shelf Area Range (m2) | Shelf Spacing and Alternative (mm | Shelf Size (w x d) (mm) | Number of Shelves and Alternative | Maximum vials loading capacity (2R vial) | Min.Condenser Capacity (kg @ 12.7 mm ice) |
| Minimax 1.0 | 1.2 | 130 | 643 x 615 x 18 mm | 3 + 1 | 4275 | 29 kg |
| Minimax 1.4 | 1.6 | 110 | 643 x 615 x 18 mm | 4 + 1 | 5700 | 29 kg |
| Minimax 2.0 | 2.4 | 130 | 643 x 930 x 18 mm | 4 + 1 | 9150 | 63 kg |
| Minimax 2.8 | 3.0 | 110 | 643 x 930 x 18 mm | 5 + 1 | 11440 | 63 kg |
| Minimax 3.3 | 3.6 | 130 | 643 x 930 x 18 mm | 6 + 1 | 13720 | 78 kg |
| Minimax 3.9 | 4.2 | 110 | 643 x 930 x 18 mm | 7 + 1 | 16000 | 78 kg |
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website

