Integrated technologies to achieve a good granulation process.
Aria fluid bed granulator and MicroNIR
Introduction
Technological advances in analytical instrumentation and computing systems have allowed many industries to better monitor and control business critical processes. This has enabled proactive, not reactive approaches to real-time quality assurance and product release. Until the advent of QbD and subsequent PAT initiatives, pharmaceutical and related industries were not fully equipped with the right guidance and supporting architecture to gain the benefits of modern, state-of-the-art quality control and assurance systems. QbD is not a single approach to the development and maintenance of a product’s lifecycle, but the utilization of many analysis and analytical tools and a modern approach to quality by subject matter experts. For many years, near infrared (NIR) spectroscopy has been the PAT tool of choice for process monitoring due to its adaptability and analytical precision. There are many applications where NIR has been utilized for process monitoring in a wide number of industrial sectors and many publications exist in the literature for the interested reader to obtain. PAT initiatives are deployed both to R&D and to real manufacturing with different targets. In the first case target is to understand the experimental domain, enabling Critical Process Parameters (CPPs) optimization depending on raw material Critical Quality Attributes (CQAs). In the latter target is to have a continuous monitoring in order to yield more reliable outputs allowing a faster and safer final product release. To apply this technology for monitoring the granulation process is essential to control the process variables that affect the granualtion process.
The cost of quality
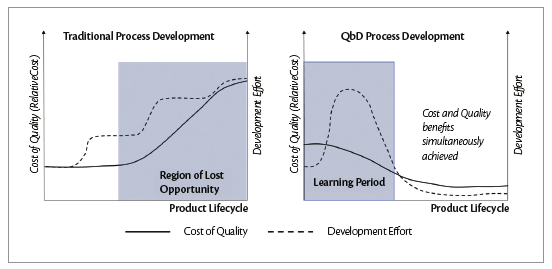
PAT deployments are connected to epochal change from traditional approach based on Quality by Test (QbT) to Quality by Design (QbD), heavily affecting in product cost. Using a traditional approach in developing of a new formulation is apparently possible to reduce development efforts but later on, due to lack of knowledge in the process, it will be time consuming undertake corrective action in case of out specifications. The process learning based on QbD, despite a longer formulation development study, allow to have more knowledge on CPPs with benefits to reduce product cost on long run. Standard communication protocol between industrial equipment called OPC DA\UA enables NIR results to be transferred on HMI for triggering data acquisition and detecting process end-point ensuring retroaction on automatic recipes for stopping the process.
Process set-up
Aria is a fluid bed processor that allows granulation, drying and pellet coating with maximum reliability and wide flexibility. Designed for through-the-wall installation, Aria is easy to integrate with other granulation equipment into fully contained processing lines.
Granulation can be defined as the size enlargement process, in which the starting material is fine powder and the final product is an aggregate in which primary particles can be still identified. The granules are held together with bonds formed by the binder used to agglomerate.
The product is suspended within the fluid bed by applying hot air. The binder liquid is sprayed onto the powder at controlled addition rate for allowing the desired particles size range. Finally, the granules are dried to evaporate the liquid and target the final residual moisture.
| 1. Raw Materials Identification (RMID) | Visual Identification | NIR handheld: real time monitoring at the point of use |
| 2. Dry Mix (Uniformity) | Fired Time | NIR collecting spectra No sampling required Achievement of desired quality attribute by trend analysis (MBSD profile) |
| 3. Binder addition (Particle Size) | Quantity of Liquid | |
| 4. Final Drying (Moisture Content) |
Fixed time |
|
| Process Recipe | Conventional Manufacturing Approach | PAT Manufacturing Approach |
Table 1: end-points used during a fluid bed granulation process.
The conventional approach to granulation process monitoring is based on physical properties (inlet and outlet air temperature and\or inlet and outlet air humidity). Despite possibility to deploy these measurements on-line, these sensors are not giving any direct measurement of particle size and moisture of the products. To better monitor the real product characteristics, sampling is required for off-line or at-line measurement, resulting in time consuming techniques for moisture content (measured by LOD or KF analysis) and particle size distribution (PSD, given by sieves or laser diffraction method). The material properties can change during the time between sampling and the availability of the results, so direct process control is not possible. On the other hand, PAT approach based on NIR sensors allowing determination of multiple parameters at the same time by measuring on-line residual moisture and particle enlargement during all phases of granulation process by achieving the desired quality attribute phase-by-phase and not at fixed time. Keeping under control the process variations, the big advantage is optimization in real time of the process parameters by reducing the risk to reject the batch. Information about process or material properties is obtained shorter than the time in which sampling properties can change. This means that on-line and in-line analysis permit continuous process control. Furthermore, the end-point determination sampling-free excludes the risk for the operator exposure to products and eliminates the analysis detection and/or human errors.
To achieve a good granulation, the starting particles must be uniformly mixed; the mixing phase is strongly influenced by the airflow rate and the air volume and particular attention is paid to the physical properties of the raw materials that have a big influence at this stage. The particle size of the resulting granule can be controlled by the quantity and the droplet size of binder liquid as well as by the rate at which it is fed.
There are a number of process variables that affect the granulation process and an accurate control of all of them is necessary for a robust system.
The MicroNIR™ PAT U spectrometer from VIAVI Solutions™ has revolutionized miniaturization of analytical instrumentation and has allowed the installation of such smart devices into process environments (including Explosive Hazardous Locations) where larger, less robust instrumentation of the past has not been able to be utilized. The MicroNIR sensors are equipped with NIR LVF filters (patented by VIAVI Solutions), without any moving parts and therefore maintenance free.
MicroNIR™ PAT U is suitable to be installed through a welded flange (sapphire glass) or in direct contact with the product (extended probe), depending on customer needs. First configuration has the advantages of no process perturbation, easy installation (eventually through a pre-existing window) and high CIP and SIP compatibility. Risk of foul with very sticky materials can be overtaken by direct contact measurements.
1. Raw materials identification
Raw materials identification is the beginning of PAT monitoring. Handheld sensors are suitable to identify and qualify all organic and inorganic substances and by allowing the loading of product and the start of production manufacturing recipe. Highest final product quality can be obtained minimizing the variability due to the flexible process based on PAT.
| Sample ID | Classified As | Results |
| batch 12345-3 | Paracetamol | OK |
| batch 12345-4 | No Match | Fail |
| batch 12345-5 | Paracetamol | OK |
| batch 12345-5 | Paracetamol | OK |
Table 2: raw materials identification.
2. Dry mix
The variability of the signals continuously recorded creates a trend analysis based on models called Moving Block Standard Deviation (MBSD) and/or Moving Block Mean (MBM) that allow the monitoring of mixing uniformity during this manufacturing phase.
The main advantage of the trend analysis approach is that there is not a need of standard to build up a model, since the trends are reflecting the spectra variation. This approach is matching particularly the need of formulators in developing new products and widely simplifying the validation documents required.
 Graphs 1 and 2: raw spectra (absorbance) and normalized pre-processed data (1st derivative) obtained by MicroNIR.
Graphs 1 and 2: raw spectra (absorbance) and normalized pre-processed data (1st derivative) obtained by MicroNIR.
3. Binder addition
Reliable in-process product characterization strictly connected with the liquid distribution onto the powders helps to adapt the process parameters accordingly to the desired quality attribute in terms of granule dimension.
NIR spectra acquired during the preliminary test carried out, are used for creating a quantitative model using a PLS (Partial Least Squares) algorithm. While NIR is acquiring continuously on-line, it is necessary to withdraw and analyze samples with wet chemistry methods. Graphs are showing outstanding correspondence between NIR predicted values and wet chemistry values for PSD (D50).

Graph 3: correspondence between NIR predicted values and wet particle size analysis.
4. Final drying
As in the dry mix phase, the MBSD profile can be used by correlating NIR MBSD (F-Test) and physical critical process parameters. The MBSD becoming constant (no variability) when the outlet air temperature, product temperature and air outlet absolute humidity tend to a plateau (Graph 4).
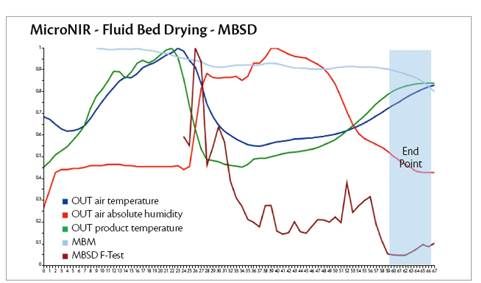 Graph 4: process parameters profile integrated with MicroNIR trend analysis.
Graph 4: process parameters profile integrated with MicroNIR trend analysis.
NIR allows to condensate all of these attributes and also it is a direct measurement of the product instead of a physical property not directly correlated to the materials dried, hence more accurate. As in binder addition phase, by using PLS algorithm is possible to correlate NIR measurement with off-line moisture content analysis (LOD%).
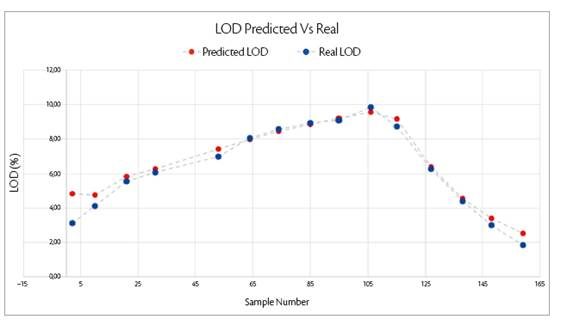 Graph 5: correspondance between NIR predicted values and moisture content analysis.
Graph 5: correspondance between NIR predicted values and moisture content analysis.
In conclusion, the integrated NIR on-line monitoring during the process offers the possibility to automatically skip at the next phase or shutting down the process once reached the target product conditions; this results in shorter production time, higher process reproducibility and easier up-scaling.
Relive
Achema
The exclusive tech videos shot during Achema 2024 are now available on our dedicated website